Engineered transparencies are an essential component of electronic devices, particularly cover screens in smartphones. Strengthened glass and, to a lesser degree, sapphire crystal have been used for these applications. Manufacturing methods for strengthened glass and sapphire bulk materials dictate costs and, to a great degree, material properties, as well as limitations of the materials for their use in consumer items.
Presumably because of its transparency in thin sheet form, sapphire screen cover often is labeled “sapphire glass” or “sapphire cover glass” in the media and in some patent applications.1 The terminology is non-scientific. Sapphire is not a glass; it is a single crystal. The fact that sapphire crystal can be “annealed” (to reduce defect concentration) also does not qualify it as a glass. Sapphire is a single crystal of aluminum oxide.
Not long ago, when we used flip phones and BlackBerry email devices, coated plastic transparencies were acceptable. They were protective, survived dropping, were moisture-proof, and were sufficiently scratch-resistant. As smart phones with uncovered screens took over, however, transparency requirements changed. They had to be protective, fracture-resistant, and scratch-resistant and had to have touch sensitivity and better optical performance.
Many manufacturers were able to meet these requirements with thin ion-exchanged, chemically strengthened glass. Corning (Corning, N.Y.), Asahi Glass (Tokyo, Japan), Nippon Electric Glass (Otsu, Japan), and Schott (Mainz, Germany) adapted known technologies to offer cover screens that are typically 0.55-mm thick, chemically strengthened, alkali aluminosilicate glass with 600–900 MPa surface compression and a compressive depth of layer of 25–60 µm.
Windows for smartphones offered by suppliers, such as Samsung (Seoul, South Korea) and Apple (Cupertino, Calif.), have an annual consumer market value of ~$5 billion.2 There is, however, a significant problem associated with these smartphones—drop-related breakage. Figure 1, adapted from Sands and Tseng,3 shows 2010 drop and spill failure rates.
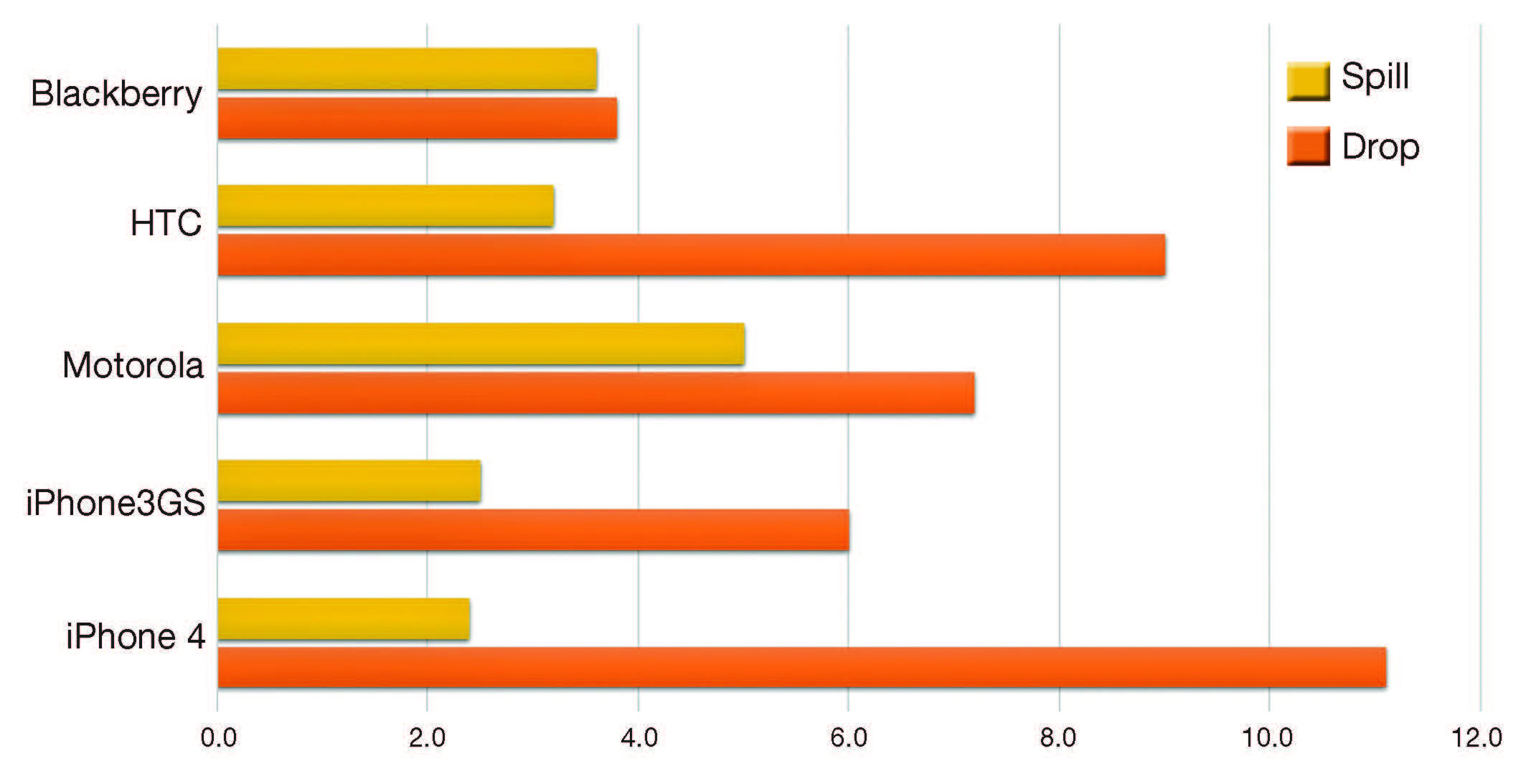
Figure 1. Incidence of smartphone drop and spill damage in first 12 months of ownership. Credit: Adapted from Austin Sands and Vince Tseng; SquareTrade Inc.
Overall drop-related failure rates are manufacturer-dependent. However, as many as ~11% of units experience drop-related damage. Drop breakage is surface- and edge-related.4 Surface breakage likely is caused by falling on a non-smooth surface, such as pavement. Edge failure likely is enhanced by device designs with exposed edges. Unfortunately, elegant, thin designs with exposed edges and without protective metal lips are most likely to fracture. With a greater emphasis on sleek looks in recent years, drop-related glass breaks appear to have increased to as high as 25% in some designs.5
Of course, glass suppliers continue to improve their material. Recently, Corning announced Gorilla Glass 5, with a claim that it will survive 80% of drops from a height of 1.6 m.6 However, those figures indicate a failure rate of ~20% from a shoulder-height drop.
Damaged glass repair is expensive, costing around $175 per screen replacement, and is highly profitable—phone vendors and authorized repair shops cite an ~90% profit margin.7
Although glass manufacturers continue to enhance their material, the current solution of necessity is protective cases and, to a much lesser extent, sacrificial screens. Protective cases have become a huge market, currently generating more than $13 billion annually at the consumer level, with a compounded annual growth rate of 5.8%.8
Sapphire is an obvious alternative material, because it has superior scratch resistance compared with any glass composition. Sapphire is a well-known cover window and often is used in watch crystals and on specialty phones, such as Vertu (Church Crookham, U.K.).9 Sapphire already is adopted on high-touchpoint smartphone areas that require extra scratch resistance, such as fingerprint sensor covers and camera lens covers.
Further, there are other advantages to sapphire. For instance, sapphire has better electrical properties, leading to increased touch sensitivity and potentially enhanced battery life. Prestige and product differentiation also are clear sapphire values. What is not clear is whether sapphire can reduce overall breakage, which will be further discussed later.
Among the negatives, cost is a major consideration for sapphire, with some figures citing a factor of 10 times the cost of Gorilla Glass.10 Certainly this gap can be reduced—but, as we review manufacturing processes, it will become evident that flat glass sheet processing is inherently much less expensive than batch, boule manufacture of sapphire.
Strengthened glass manufacturing
Thin flat glass is made using one of four techniques: the Corning fusion overflow process; the Asahi float method; Schott’s down-draw process; or the now obsolete process of grinding down and polishing a thicker plate.
Corning’s fusion overflow is the most elegant method, in which fused glass overflows a trough or pipe on both sides. The overflowing layers are brought together at the bottom and drawn into a very thin, flat, uniform-thickness sheet with two pristine, equivalent surfaces (Figure 2).11 Alkali aluminosilicate compositions are marketed by Corning as Gorilla Glass.
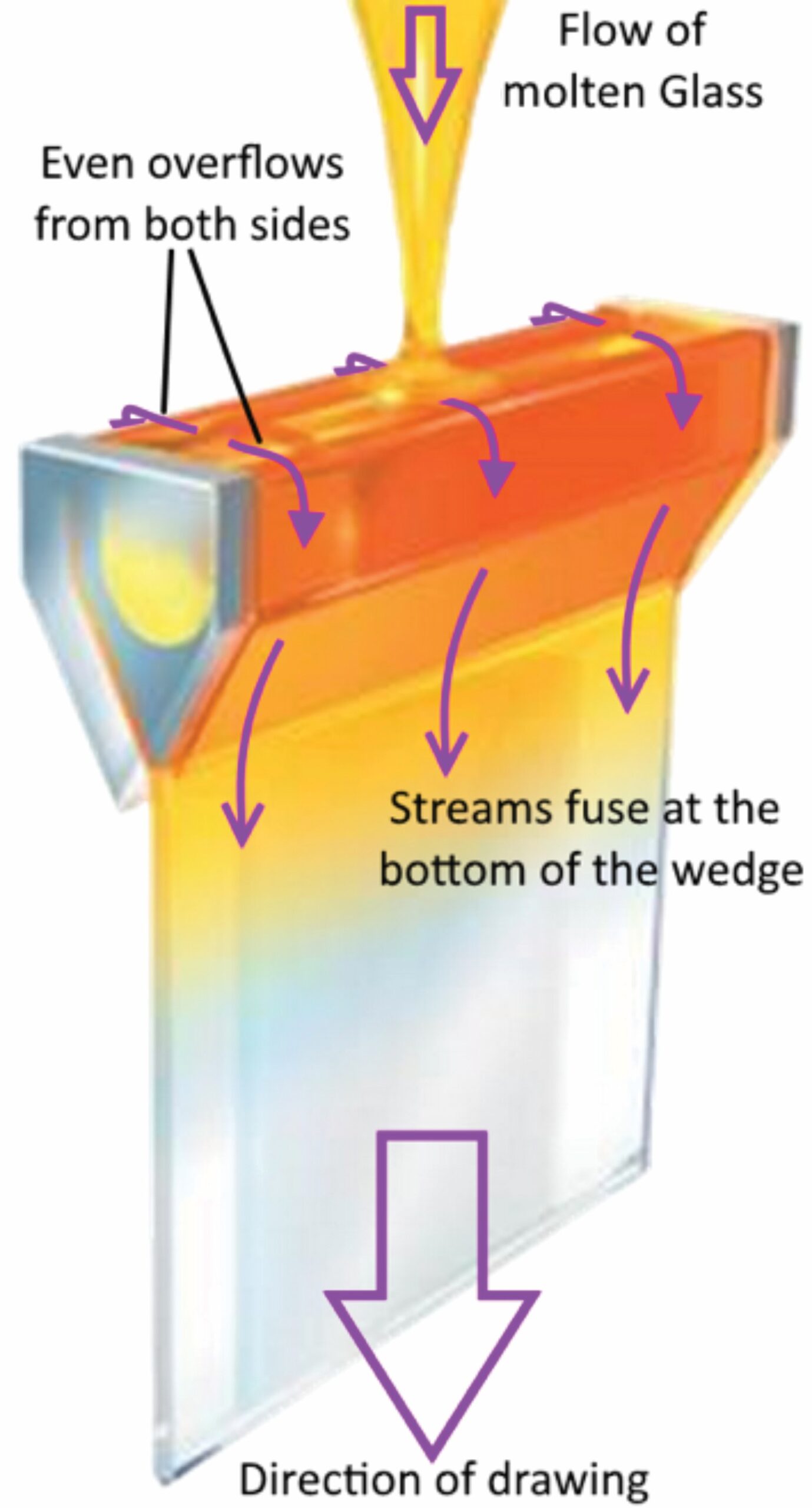
Figure 2. Corning fusion overflow process for producing thin glass (adapted from corning.com). Credit: Corning Inc.; Arun Varshneya
Pilkington of the United Kingdom, now part of Nippon Sheet Glass, invented the float process in the early 1950s.12 Molten glass flows horizontally over a bath of molten tin, entering around the working point viscosity and exiting in a relatively solid ribbon form. Under interfacial surface forces with tin and its gravity, silicate glasses usually acquire an equilibrium thickness of ~6 mm. To make it thin, the glass is stretched away from the center using top rolls.
Using the float process, Asahi produces Dragontrail glass, which is an alkali aluminoborosilicate with a fairly high glass transition temperature (Tg). Relative to Corning fusion overflow, the float process can draw thinner glasses, but the big advantage is size—the process can generate sheets as big as 3 m wide. The relatively slower cooling rate through the glass transition additionally produces a more stable glass. The two surfaces of a float-produced glass, however, are not equivalent. The surface in contact with molten tin (“tin surface”) does show substantial tin absorption, as deep as 2–10 µm. The upper surface (“air surface”) has significantly less tin absorption from the atmosphere in the melting chamber.
Beyond forming, glass sheets need to be cut—generally using a laser, water jet, or mechanical wheel—beveled with CNC machines, drilled with holes, polished on the edges, and then chemically strengthened.
Chemical strengthening is now a well-understood process.13,14 Briefly, glass containing alkali ions, such as Na+, is immersed in a bath of molten KNO3 salt at temperatures just below the strain point. With time, Na+ ions migrate out of the glass, and their place is taken up by relatively larger K+ ions stuffed into the structure on a one-to-one basis (Figure 3). The stuffing produces large surface compression, which must be overcome by an applied tensile stress before glass can reach its strength to fracture. Then, glass is effectively strengthened. In addition, the process also increases scratch resistance.
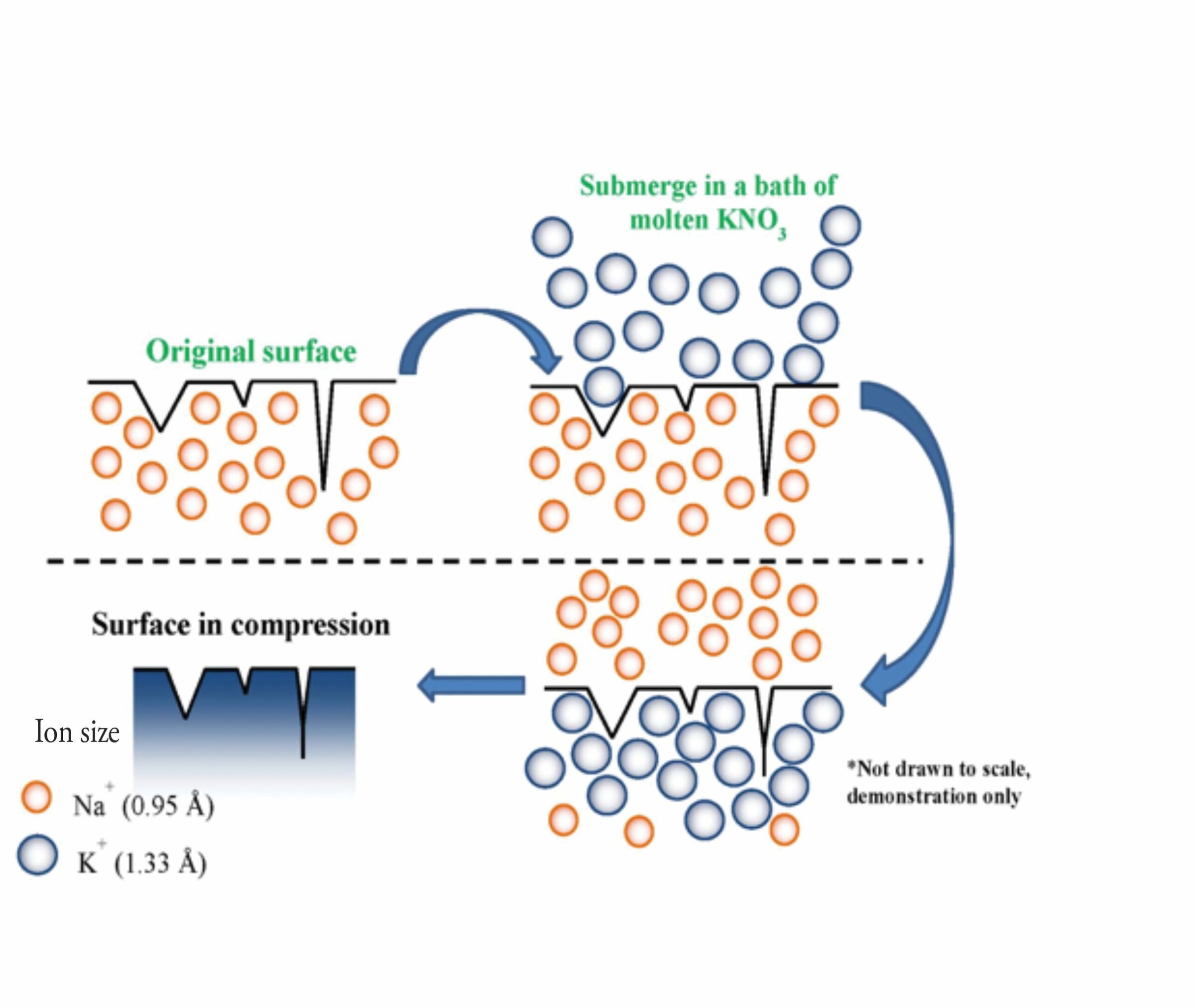
Figure 3. Chemical strengthening fundamentals. Credit: Arun Varshneya
The major advantages of chemical strengthening over traditional thermal tempering are
- A large protective surface compression magnitude (for example, Gorilla Glass and Dragontrail are capable of generating 800–900 MPa);
- Glass as thin as 25 µm can be chemically strengthened;
- Being an immersion process, curved glasses can be strengthened just as readily as flat ones; and
- After-process optics for through-vision do not show significant deformation, because processing is below the Tg.
Disadvantages include
- A relatively low depth of layer of only a few micrometers, just barely greater than the usual handling flaws but nowhere near to what an indenting projectile could penetrate; and
- Rather high costs associated with hours of salt immersion.
However, the high cost of ion exchange-strengthened glass relative to the thermal tempering process remains a bargain with respect to sapphire processing.
Float-produced glasses, although less expensive than those produced using the fusion overflow process, suffer from warping after chemical strengthening, because varying amounts of tin on the two surfaces result in unequal alkali-ion interdiffusion. For screens thinner than ~1 mm, manufacturers must adopt some method to reduce warp during chemical strengthening.
Sapphire manufacturing
Sapphire is a crystal with a melting point of 2,044°C. During the past century, many commercial manufacturing approaches have been developed for sapphire. The flame fusion approach, attributed to the Parisian Auguste Verneuil, has origins in a Geneva process for making synthetic rubies.15 This process uses a vertically down-directed oxyhydrogen torch capable of flame-melting finely ground purified alumina.
The relatively simple and inexpensive process—in which alumina powder is injected into the flame, fused, and deposited and recrystallized on a rotating target containing a sapphire seed—continues to be used. The process typically produces relatively small synthetic gemstones as well as industrial products, such as watch crystals. Such synthetic stones often are acceptable aesthetically, although they have striations or growth rings—a result of target rotation, which presents a leading and trailing thermal edge to material deposition.
The Czochralski (CZ) process is another well-known method, especially for production of silicon, since its accidental discovery by Jan Czochralski in 1916.16 As practiced today, the process dips a rotating, oriented alumina crystalline seed into a counter-rotating crucible of molten material contained within an inert atmosphere furnace. A single cylindrical crystal with a predetermined orientation is withdrawn. Although size limited and relatively expensive, the CZ process is typically used for producing optical quality sapphire for electronic applications.
The edge-defined film growth (EFG) process, developed by Tyco Industries in 1965,17 can be viewed as a variation on the CZ technique. The process uses a die that directly forms the desired near net shape (Figure 4) and can directly form tubes, rods, or ribbons, minimizing subsequent machine time, costs, and material consumption. Although minimizing costs, this approach produces a relatively low optical quality product, which often is used for industrial-grade optical and mechanical applications.
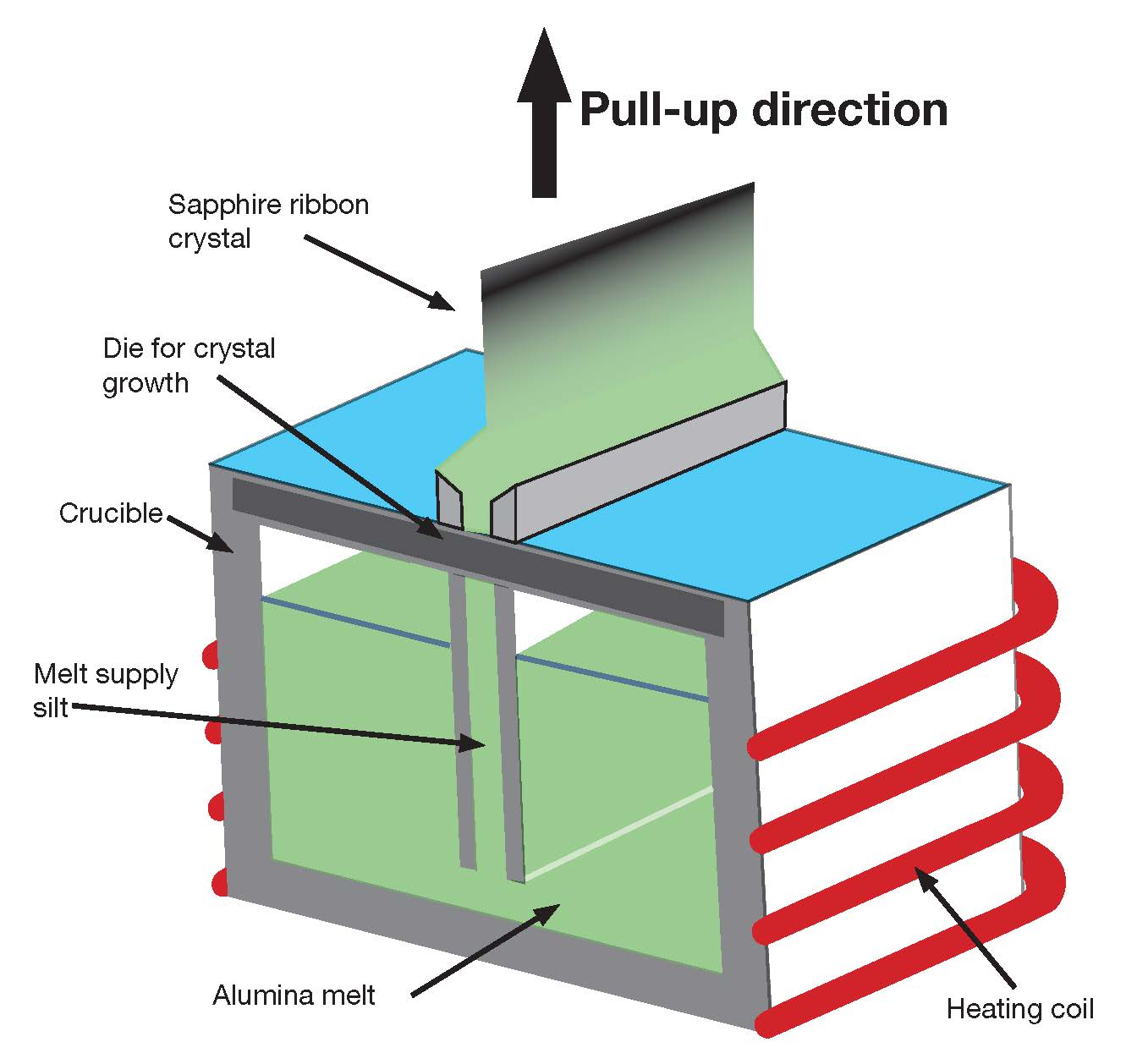
Figure 4. The edge-defined film growth process, a variation of the Czochralski process, uses a die to directly form a near-net shape. Credit: Adapted from substech.com
Large-boule techniques are most amenable to minimizing cost while achieving satisfactory optical quality. To address size limitations of the Verneuil and CZ approaches, Spyro Kyropoulos18 developed a direct melt process in 1926. In this method, raw material, alumina powder, and crackle are melted in a refractory—typically molybdenum—crucible. A seed is inserted to initiate crystallization and is slowly withdrawn, growing a controlled crystal from the crystal–molten interface through careful thermal gradient control (Figure 5). This process results in a relatively large, stress-free boule with high optical quality. Subsequent core drilling along any chosen crystallographic axis can create a variety of optical and electronic windows, lenses, and substrates.
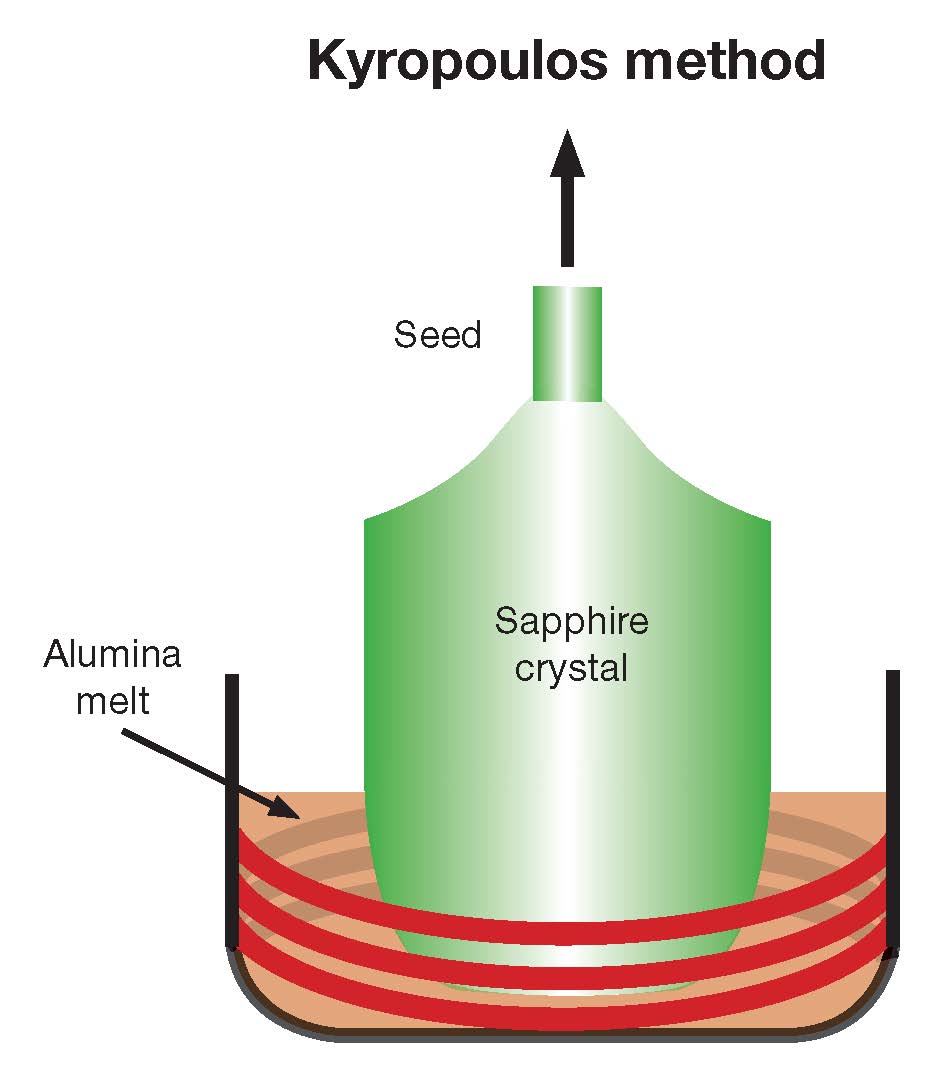
Figure 5. The Kyropoulos process uses a seed to initiate crystallization from raw molten material and produces a relatively large, stress-free boule with high optical quality. Credit: Adapted from substech.com
More recently, GT Advanced Technologies (GTAT, Merrimack, N.H.) used an advanced sapphire furnace19 in a variation on the heat exchange method, originally developed at the former Crystal Systems in 1967.20 The heat exchange method itself is a variation on the Kyropoulos method. In the advanced sapphire furnace approach, a refractory crucible in an evacuated furnace, with a seed at the bottom, is charged with high-purity alumina raw material. The charge is melted, and a cold finger in thermal contact with the seed withdraws heat, preventing the seed from melting. The seed initiates crystallization of the melt. Careful thermal control results in a controlled fusion front traveling from the bottom (Figure 6), resulting in a very large, optical quality boule. In practice, as GTAT found, managing stress and achieving crack-free boules can, ultimately, be size-limiting.
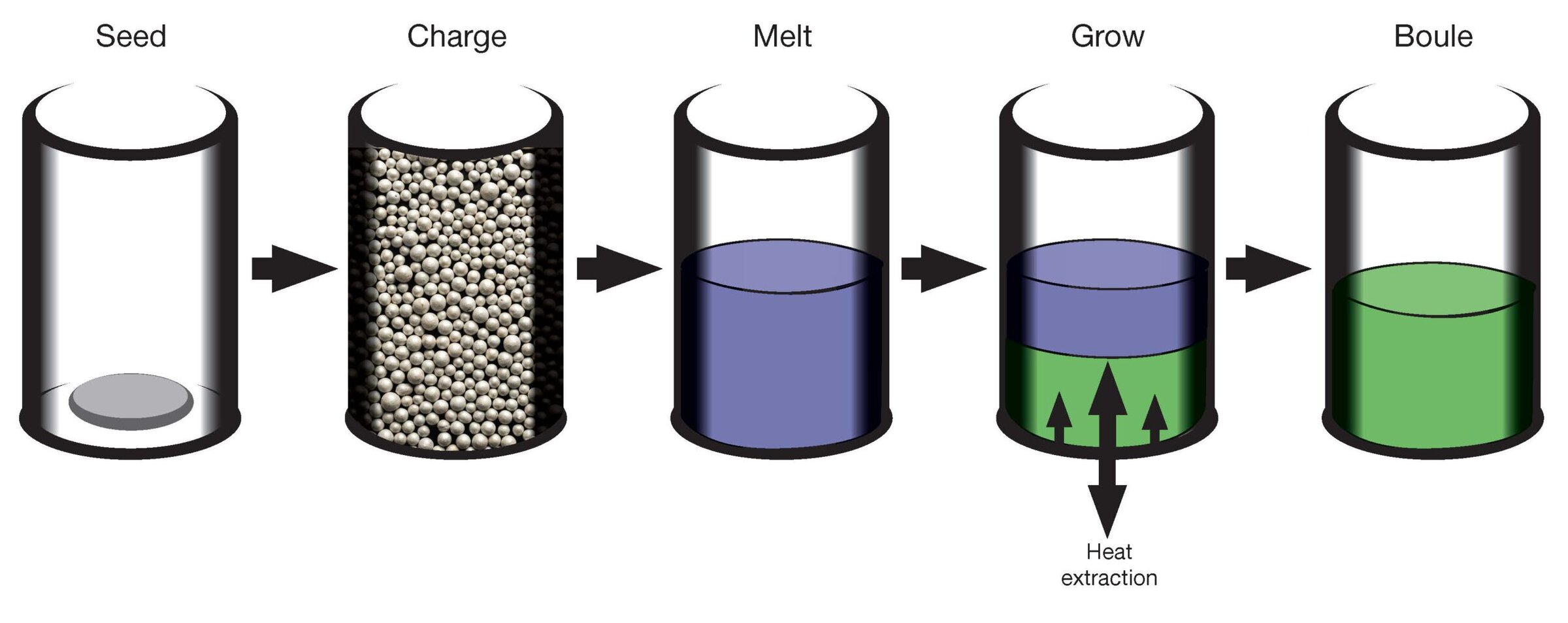
Figure 6. GTAT’s advanced sapphire furnace approach uses careful thermal control to generate a controlled fusion front traveling from the bottom, resulting in a large, optical quality boule. Credit: Adapted from J. Zahler, “Sapphire and GaN Substrate Materials,” 2012 DOE SSL Manufacturing R&D Workshop.
Sapphire boules need to be thin-sliced into wafers, which generates substantial kerf loss. The thin slices are drilled for appropriate slots, edged or beveled, and mechanically polished. A postprocess annealing step at ~1,800°C substantially helps reduce strength-degrading effects of mechanical damage on the surface.21 Following bulk material manufacture, article fabrication and device making (including application of surface coatings) are necessary to produce a finished transparent cover plate. Screen fabrication processes are energy intensive, often as high as ~50% relative to boule-making energy.
In October 2013, GTAT contracted22 with Apple to supply sapphire raw material as a proposed glass replacement. However, GTAT filed for bankruptcy in October 2014. The company failed for a variety of commercial reasons, but fundamentally, it could not meet quality and target economics—the technical-limiting factor was cracks.
Property comparison
An electronic display cover window should have good scratch resistance, low ambient light glare, good touch sensitivity, and, as we have stressed, not break when dropped (i.e., high fracture toughness).
Table 1 summarizes the relevant physical properties for sapphire and several commercially available chemically strengthened glasses. Briefly, sapphire has three times the fracture toughness (KIc) and three times the hardness, but is significantly heavier with a higher refractive index and dielectric constant than its strengthened-glass competitors. Although lighter is better, weight difference is not a major concern for smartphone cover screens. Fracture toughness and scratch resistance are more important parameters, and strengthened glasses are no match for sapphire. Sapphire also has a higher dielectric constant, which potentially enhances touch sensitivity and battery performance.
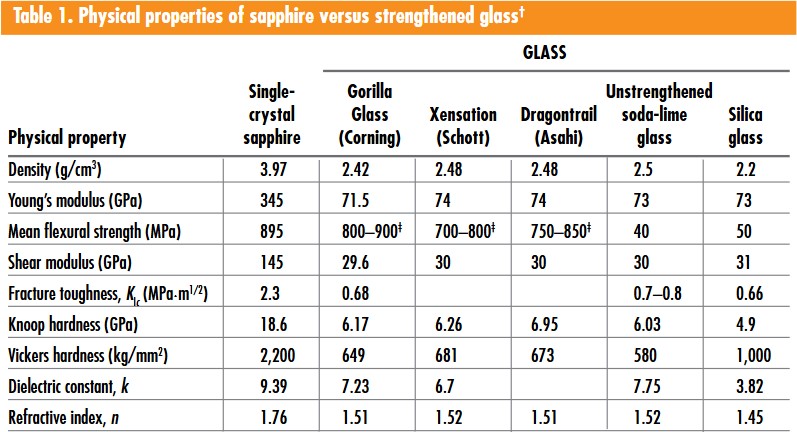
†Adapted from GTAT, www.gtat.com, “GT ASF Grown Sapphire Cover and Touch Screen Material.”
‡Estimated by the authors.
Sapphire also has five times higher Young’s modulus of elasticity than glass. This implies that, given a certain application of flexure force, sapphire has considerably less strain. This also means that the liquid crystal display (LCD) behind the sapphire screen suffers much less distortion, providing superior and longer performance.
Sapphire has a higher refractive index of 1.76, in comparison with 1.51 for glass—this corresponds to approximately 1.8-times greater reflection. In the dark, the two covers appear similar. However, in ambient light, sapphire cover displays appear significantly diminished.
Aesthetics of design versus material performance capability
Over the years, product aesthetics and ergonomics of holding the device have taken a dominant role.23 Designing to win market admiration includes 2.5-D, where the screen is slightly curved at the edge, and 3-D, where the screen is bent to cover the edge. Whereas it is possible to make 2.5-D screens out of sapphire using very special tooling to conduct material removal and polishing, 3-D is extremely difficult and expensive, if not impossible. Glass has a clear advantage in these designs, and this is possibly the main reason for its preference over sapphire. Of course, design trends can and will change—consumers may become dissatisfied with ultrathin, fragile devices that require frequent fixing or replacing.
Product drop breakage performance can be more complicated and harder to predict. It is a consequence of device design and details of the impact that initiates failure. Sapphire may have a significant advantage over glass because of its high fracture toughness and high hardness. According to Heuer24 and Raghvan,25 the flexural strength and presumably impact resistance of sapphire are most dependent upon quality of polish and quality of annealing, which control the surface and subsurface flaw density generated by machining damage.
Laboratory strength tests can be selected to determine advantageous results and are not necessarily good predictors of actual device performance. Glass screens might well survive flexure-limited strength testing better than sapphire. However, scratched glass will be far less likely to survive a drop in actual use. Further, all of this will be aggravated by exposed edge designs. In actual use over a longer time period, greater cumulated scratches in strengthened glass are likely to make it the weaker screen material. Nonetheless, we are not aware of statistically significant field or simulated usage data that prove or disprove this hypothesis.
Cost comparison
Less controversial is the strengthened glass cost advantage. Calculations (Corning and others)10 have shown, not surprisingly, that sapphire costs significantly more to manufacture and requires more energy to do so. Estimates of $3 per glass cover plate are approximately one tenth that of a finished sapphire plate. Energy is a significant part of this cost difference—sapphire consumes up to 100 times the energy necessary to make Gorilla Glass.10 In addition, 2.5-D designs for a sapphire screen can increase costs significantly. However, cost and manufacturing energy requirement differences certainly can be somewhat mitigated with future process improvements.
Future trends
Future improvements are likely to be in four directions: design, glass, sapphire, and alternate materials.
Because drops on smartphone edges are a major issue, design-incorporated edge protection would substantially reduce screen damage.
Float glass warp after chemical strengthening can be reduced simply by polishing away the “tin side” prior to chemical strengthening. Newer and perhaps more economical methods are to use one of several techniques26-29 that conduct differentially augmented strengthening on the “tin side.” For example, in the “differential density” method, one coats the air surface with a thinner layer of a paste-based salt relative to the tinned surface, thus allowing differential areal uptake of potassium to reduce surface warp.26
The use of stress relaxation control also can generate higher surface compression with comparable depth of layer.14 An example is Saxon Glass’s High-CS Ionex glass,30 in which controlled additives in the salt bath develops as much as 40–100 MPa higher surface compression. Certain ions lodge themselves into the glass surface or enter the network, where they impede network relaxation.
Another possibility is to use electric field-assisted chemical strengthening. Because field-assisted ion exchange is conducted over a much shorter time relative to traditional exchange, the surfaces experience significantly less stress relaxation, which generates a much higher value of surface compression.14 An example of such a product is Saxon Glass’s Frankenstein, in which a multicycling direct-current potential assists development of a fast ion-exchanged layer with stress balance and an advantageous flatter compressive stress profile.31
For sapphire to be a realistic challenger to glass, a major improvement would be for the technology to develop 1.5–2.0 GPa strength (in ring-on-ring or four-point bend strength measurements). There is evidence to suggest that more careful annealing during boule growth may reduce thermal stresses, which otherwise cause strength-degrading deformation twin misfit boundaries.21 Kerf reduction and layout optimization, such as the potential for square boules, will help with material utilization. In short, there is a clear need for quality and growth-rate improvements in sapphire processes, including nonboule or near-net-shape and wafer fabrication processes.
Finally, alternate materials and technologies also are worth pursuing. For glass, a chemically strengthened transparent glass-ceramic32,33 may have the needed benefits of somewhat higher fracture toughness (~1.1 MPa∙m1/2) and higher strength. Except for an additional ceramming step, glass-ceramics enjoy all of the fabrication ease of traditional glasses. For transparent ceramics, zirconia and spinel crystalline ceramics may be good candidates.
A final comparison
Manufacturing glass substrates consumes significantly less energy than large scale sapphire manufacturing processes. Therefore, glass has considerable fabrication cost advantages in addition to greater transparency for applications, such as cover screens in personal mobile electronics. However, scratch resistance and breakage caused by accidental drops remain an issue for glass. Although sapphire has much higher scratch resistance and fracture toughness, its strength remains questionable because of machining damage and crystal defects.
Consumer preference for aesthetics in an “all-glass” design with exposed edges and faces has played a key role in the dominance of glass so far. GTAT’s recent failure to manufacture and supply sapphire transparencies leaves chemically strengthened glass as the only volume scale cover glass option. Customers will continue to purchase protective cases and pay for replacing cracked screen covers. For crystalline ceramics to be competitive, quality improvements and large cost reductions in fabrication are imperative. Alternative materials, like chemically strengthened glass-ceramics, may also have a future in this market.
Capsule summary
Consumer preferences drive material choices
As smart phones with uncovered transparent touchscreens have dominated consumer preferences, glass and sapphire materials have taken over from coated plastic transparencies. But which is more advantageous—strengthened glass or sapphire—and what is driving the future of new materials for cover screens?
Reviewing the choices
We analyze and compare material manufacturing and article fabrication processes for strengthened glass and sapphire crystal and address advantages and disadvantages for physical properties, such as strength, abrasion resistance, fracture toughness, light reflection, and touch sensing, for these applications.
The future is clear
Although glass has considerable fabrication cost advantages and greater transparency, sapphire has much higher scratch resistance and fracture toughness for cover screens in personal mobile electronics. Nonetheless, consumer aesthetic preferences and manufacturing ease will continue to dictate existing or novel material preferences for cover screens.
Cite this article
A. K. Varshneya and P. P. Bihuniak, “Cover screens for personal electronic devices: Strengthened glass or sapphire?” Am. Ceram. Soc. Bull. 2017, 96(5): 20–25.
About the Author(s)
Arun K. Varshneya is a Distinguished Life Member of ACerS, president of Saxon Glass Technologies Inc. (Alfred, N.Y.), and Professor of Glass Science & Engineering, Emeritus, Alfred University. Peter P. Bihuniak is a Fellow of ACerS and president of Hidden Point Consulting LLC (Chagrin Falls, Ohio), and Advisory Board Partner with X-Roads Partners. Formerly he was CTO of GT Solar. Contact Varshneya at varshneya@alfred.edu.
Issue
Category
- Electronics
- Glass and optical materials
Article References
1K. Kwong, “Laminated aluminum oxide cover component,” U.S. Pat. Appl., US 2014/0193606A1, July 10, 2014.; See also patent applications US2014/0139978A1; US2014/0138221A1; and US2014/192467A1.
2“Smartphones and tablets still drive demand for cover glass, as the industry looks to smart watches for growth, IHS Says,” IHS Markit, July 9, 2015.
3A. Sands and V. Tseng, Square Trade Research Brief, “Smart Phone reliability: Apple iPhones with fewest failures, and major Android manufacturers not far behind”, Nov 3, 2010.
4AGC Dragontrail X brochure, http://dragontrail.agc.com/en/.
5M. Wilson, Co.Design, “Why Won’t apple Fix the iPhone’s One Huge Flaw?”, Aug. 30, 2016.
6T. Maddox, “Gorilla Glass 5 will protect phones and tablets from 80% of drops,” TechRepublic, July 21, 2016.
7M. Asay, “The ridiculous profit made form replacing cracked smartphone screens,” TechRepublic, Sept. 2, 2015.
8“Mobile phone protective cases market,” Report number 58085-09-16, September 2016. http://www.credenceresearch.com/report/mobile-phone-protective-cases-market
10S. Hill, “Glass warfare: Why unscratchable sapphire screens have Corning beating its chest,” Digital Trends, March 12, 2016.
11S.M. Dockerty, “Sheet forming apparatus,” U.S. Pat. No. 3 338 696, 1967.
12L.A.B. Pilkington, “Review lecture: The float glass process.” Proc. R. Soc. London, 314, 1–25 (1969) doi : 10.1098/rspa.1969.0212.
13A.K. Varshneya, “Chemical strengthening of glass: Lessons learned and yet to be learned,” Int. J. Appl. Glass Sci., 1 [2] 131–42 (2010).
14A.K. Varshneya, G.A. Olson, P.K. Kreski, and P.K. Gupta, “Buildup and relaxation of stress in chemically strengthened glass,” J. Non-Cryst. Solids, 427, 91–97(2015).
15Verneuil process, http://www.clearlysapphire.com/Growth.
16Czochralski process, http://www.clearlysapphire.com/Growth.
17Edge-defined film growth method, http://www.clearlysapphire.com/Growth.
18Kyropoulos method, http://www.clearlysapphire.com/Growth.
19Advanced sapphire furnace process: J. Zahler, “Sapphire and GaN substrate materials,” 2012 DOE SSL Manufacturing R&D Workshop, San Jose, Calif., June 15, 2012; slide 8.
20Heat exchange method: D.C. Harris, “A century of sapphire crystal growth”; pp. 10–12 in Proceedings of the 10th DoD Electromagnetic Windows Symposium (Norfolk, Va., May 2004).
21(a) A.H. Heuer, “The influence of annealing on the strength of corundum crystals,” Proc. Br. Ceram. Soc., 6, 17–27 (1966). (b) H. Platus, R.P. Welle, and P.M. Adams, “Sapphire window laser edge annealing,” U.S. Pat. No. 5 697 998, Dec. 16, 1997.
22B. Sanders, “GTAT-Apple agreement: Evolution of a fiasco,” NH Business Review, Dec. 11, 2014.
23K. Campbell-Dollaghan, “Apple’s strange obsession with fragility. Apple’s fine-print warning about the jet-black iPhone 7: Use a case,” 3 Minute Read, Sept. 8, 2016.
24A.H. Heuer, conversation, Nov. 2016.
25S. Raghavan, GT, conversation, Sept. 21, 2016.
26P.K. Kreski, “Strengthened glass and methods for making using differential density,” US 9,302,938 April 05, 2016.
27P.K. Kreski, “Strengthened glass and methods for making using differential time,” US2014/0178689 June 26, 2014.
28P.K. Kreski, “Strengthened glass and methods for making using differential chemistry,” US2014/0178691 June 26, 2014.
29A. K. Varshneya and P.K. Kreski, “Strengthened glass and methods for making using heat treatment,” US2014/0178663 June 26, 2014.
30High-CS Ionex, www.saxonglass.com
31A.K. Varshneya, G.A. Olson, and P.K. Kreski, “Strengthened glass and methods for making utilizing electrical field assist,” US2015/0166407 June 18, 2015.
32G.H. Beall, B.R. Karstetter, and H.L. Rittler, “Crystallization and chemical strengthening of stuffed b-quartz glass-ceramics,” J. Am. Ceram. Soc., 50 [4] 181–90 (1967).
33G.H. Beall, M. Comte, M.J. Dejneka, P. Marques, P. Pradeau, and C. Smith, “Ion-exchange in glass-ceramics,” Front. Mater., 3:41, Aug. 23, 2016, doi:10.3389/fmats.2016.00041
Related Articles
Market Insights
Engineered ceramics support the past, present, and future of aerospace ambitions
Engineered ceramics play key roles in aerospace applications, from structural components to protective coatings that can withstand the high-temperature, reactive environments. Perhaps the earliest success of ceramics in aerospace applications was the use of yttria-stabilized zirconia (YSZ) as thermal barrier coatings (TBCs) on nickel-based superalloys for turbine engine applications. These…
Market Insights
Aerospace ceramics: Global markets to 2029
The global market for aerospace ceramics was valued at $5.3 billion in 2023 and is expected to grow at a compound annual growth rate (CAGR) of 8.0% to reach $8.2 billion by the end of 2029. According to the International Energy Agency, the aviation industry was responsible for 2.5% of…
Market Insights
Innovations in access and technology secure clean water around the world
Food, water, and shelter—the basic necessities of life—are scarce for millions of people around the world. Yet even when these resources are technically obtainable, they may not be available in a format that supports healthy living. Approximately 115 million people worldwide depend on untreated surface water for their daily needs,…





