Concepts of “heat” and “temperature” pervade our thinking and language. We know intuitively that “hot” is fast and “cold” is slow. A “hot topic” also can be “cool.” We live in a narrow range of temperatures between the freezing and boiling points of water. Since early in human history, we have endeavored to harness fire to inhabit a comfortable temperature environment, to cook our food, and to make new metal and ceramic materials using high-temperature processing.

A laser-heated sample being dropped into a calorimeter.
Yet an exact definition of “high-temperature” remains elusive. The temperature of the sun’s surface and of the boundary of the earth’s iron–nickel inner core is around 5,500°C. Temperatures of millions of degrees are achieved in stars, in nuclear explosions, and in experiments on inertial confinement fusion. Temperatures above the melting and softening points of steel and glass represent high-temperature for most people, and traditionally the term ‘refractories’ is reserved for materials that can be used for containment of metal and silicate melts.
The calculation of phase diagrams method (CalPhaD)1 was developed from endeavors in alloy modeling and now is a cornerstone of the integrated computational materials engineering (ICME) approach, providing inputs for simulating phase and microstructure evolution and mechanical properties with phase field techniques and finite-element methods. ICME has demonstrated remarkable achievements in accelerated development of new alloys.2 However, with superalloys reaching their high-temperature limits for required applications,3 strong impetus exists to extend ICME to ceramic materials, which require experimentally measured high-temperature thermodynamic properties. As with any technology, the applications are stretching beyond the original development purpose. However, once successfully implemented, ICME has potential to affect all categories of refractory ceramics, as shown in Figure 1 and described briefly below.
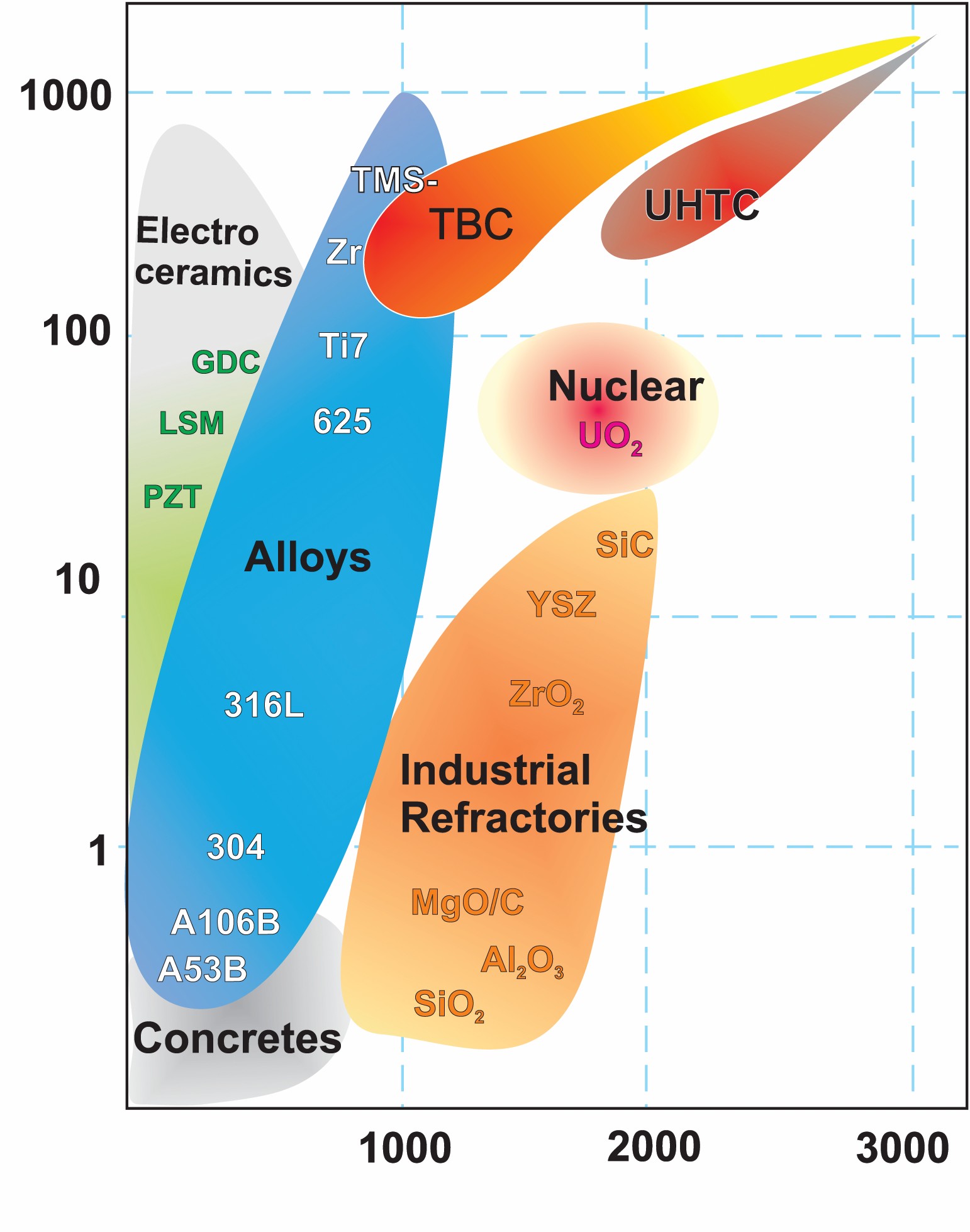
Figure 1. Operation temperature versus cost for major categories of ceramics and alloys. Credit: Alexandra Navrotsky and Sergey V. Ushakov
Major classes of refractories
Ceramic thermal barrier coatings (TBC) on metal alloys enable higher operating temperatures and prevent melting and high-temperature oxidation. In addition to high melting temperatures, TBCs are required to have low thermal conductivity and low oxygen permeability. However, lower thermal expansion of ceramic materials than of metals poses a problem of delamination during heating–cooling cycles.
Coatings for gas-turbine blades in jet engines are probably the most studied class of TBCs.3 Current coatings use tetragonal yttria-stabilized zirconia (YSZ) deposited in a columnar structure to avoid delamination. In addition, an engineered Al2O3-based bonding layer between the alloy and coating provides adhesion. Coatings with lower thermal conductivities at high temperatures, such as La2Zr2O7, have been explored4 to increase operating temperature and, thus, improve engine efficiency. Resilience of the coatings to interaction with silicate particles in the atmosphere or from the ground during takeoff and landing is a major concern and constitutes another research direction. Radiation cooling is the most efficient way to dissipate heat at high temperatures, thus, high emissivity is highly desirable.
Ultra-high-temperature ceramics (UHTCs) are under development for control- and propulsion-related surfaces of hypersonic vehicles.3 An ideal UHTC is lightweight and has a high melting temperature. SiC composites have been used for blunt aerodynamic surfaces of space shuttles, which heat to 1,500°C upon atmospheric re-entry. Sharp leading edges can provide better aerodynamic characteristics, although they can reach temperatures in excess of 2,000°C. Therefore, carbides and borides of hafnium, tantalum, and zirconium—with melting temperatures that exceed 3,000°C—are key constituents for new UHTC ceramics and impose requirements different from coatings. As with coatings on metals, their function is to protect underlying ceramics from oxidation by air or high-temperature water vapor formed during fuel combustion. Melting temperatures of constituents of UHTCs are higher than for oxides (Figure 2). For internal surfaces that cannot be cooled by radiation, high thermal conductivity may be desired to reduce thermal gradients and allow heat dissipation in other parts of the vehicle.
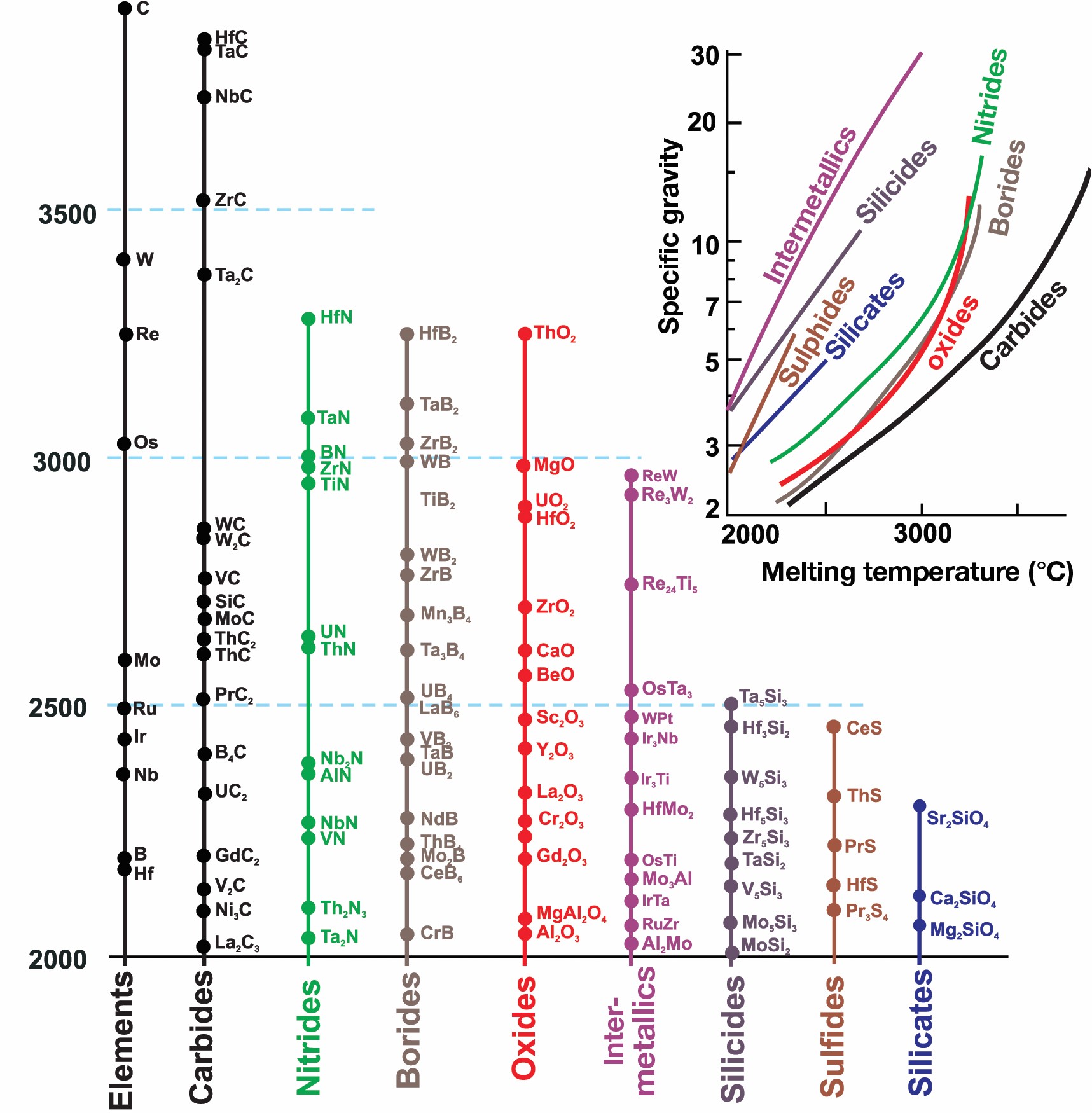
Figure 2. Compounds with melting temperatures higher than 2,000°C and specific gravity–melting temperature envelope.7 Credit: Alexandra Navrotsky and Sergey V. Ushakov
Uranium oxide (UO2) is the main nuclear fuel used throughout the world. Researchers have spent hundreds of person-years studying its high-temperature thermophysical properties and have developed many novel experimental techniques. In pressurized and boiling water reactors, UO2 is used in the form of pellets that are ~1 cm in diameter inside zircaloy tubes filled with helium gas. Composition and microstructure of the fuel changes during burnup because new atomic species are formed by fission and because of the steep thermal gradient during operation (from 1,400°C in the center of the pellet to 400°C at the rim). Reaction of zirconium with steam at temperatures greater than 1,200°C to produce zirconia and hydrogen has been the source of catastrophic explosions in nuclear reactor accidents involving loss of coolant.
There is now great interest in finding new fuels and fuel assemblies that are more accident tolerant, including uranium carbides and nitrides. Multilayer encapsulation of fuel spheres in layers of materials with high thermal conductivity such as SiC and graphite, is proposed to aid heat exchange and provide better stability in operation and direct disposal of spent fuel. ICME application to nuclear fuels is the most advanced, with an extensive and growing set of experimental data and ab initio calculations on thermodynamic and thermophysical properties of UO2 and its solid solutions, facilitating CalPhaD1 and phase field2 modeling.
Concrete and industrial refractories used for metal and glass manufacturing to a large extent use as-mined, chemically unprocessed, and relatively cheap mixtures. Thermal stability and mechanical properties of the concrete are defined mostly by cements holding together sand, gravel, and crushed rock. Portland cements, the most common type used around the world, are mostly composed of Ca2SiO5 and Ca2SiO4 synthesized at temperatures higher than 1,400°C. Calcium aluminate cements are used in construction concretes and in castable refractories, blurring the line between these categories. Oxides of aluminum, silicon, magnesium, calcium, titanium, zirconium, and chromium, and carbides and nitrides of titanium, boron, and silicon are the major constituents of common refractories. Further, oxide/graphite refractories are used to avoid wetting by silicate slags and to increase thermal conductivity.
Refractories used to be disposed as landfill after removal from service, so the cost to produce refractories was a major concern in design and manufacturing. Energy conservation and recycling requirements as well as developments in hot repair and in situ fabrication technologies, may lead to feasibility of using high-performance, more expensive formulations. Industrial refractories used at high temperatures require strength, dimensional stability, and high or low thermal conductivity, with evolving microstructure under conditions of sharp thermal gradients and in contact with silicate slags and metal melts of complex compositions.
Modeling capabilities promise to streamline development of new high-temperature materials and facilitate improvements of existing ones by reducing the cost and time required for screening and by focusing experimental efforts in the most promising directions. Data on surface energies, enthalpy, heat capacity, and molar volumes as a function of temperature are required as thermodynamic inputs. Success of the ICME approach for alloys development2 is to a large extent because of availability of these data for metals, leveraged by application of CalPhaD modeling. Fusion enthalpies have been measured for all metals and many alloys, often using various experimental techniques.5,6
However, even melting temperatures of key refractory oxides are being corrected by up to hundreds of degrees (e.g., MgO),7 heats of fusion are often unknown, and thermodynamic data for complex ternary and multicomponent oxide systems are far less complete than for metals. What follows are some examples of recent developments in experimental methods that can provide missing data. More detailed accounts of the research mentioned can be found in several reviews7–9 and the references therein.
Temperature measurement
Resistance and thermocouple thermometry is well developed and used ubiquitously. Seebeck voltage is standardized to 1,820°C for B-type Pt-Rh thermocouples, and W-Re thermocouples can be used to 2,800°C in inert conditions. Reaction with measured samples, aging, and evaporation of thermocouples makes contactless temperature measurement preferable for many applications. Often it is the only feasible method available, such as for oxidizing conditions in excess of 1,800°C, liquid metals, or levitated samples.
Radiation thermometry is used to define the International Temperature Scale above the freezing temperature of silver (961.78°C). The theoretical basis for radiation thermometry is Planck’s law describing the temperature dependence of spectral radiance of a blackbody, namely an abstract object that does not reflect light and emits only thermal radiation:
Bv(T) = 2hv3/c2) / (exp(hv/kT)–1)
where T is absolute temperature; v frequency of radiation; and h, k, and c constants (Planck’s, Boltzmann’s, and speed of light, respectively).
Emissivity (ε) is the ratio of spectral radiance of the actual material to radiance of a blackbody at the same temperature. In traditional single-color pyrometry, apparent temperature is obtained from a single intensity value registered on a narrow preselected band (ensured by a red filter in disappearing filament pyrometry). Geometry close to that of a blackbody cavity is sought in the experimental arrangement to ensure ε ≈ 1.
In the simplest case, the pyrometer can be aimed inside the crucible through a small orifice. Even in these ideal conditions, researchers must account for absorption by windows along the observation path, and any changes caused by absorption or emission, such as by generated vapors, will affect accuracy of the measurements.
When blackbody cavity geometry is not possible, emissivity has to be measured or estimated for single-band pyrometry. In general, ε changes with wavelength, temperature, observation angle, and surface roughness, and, thus, estimation of emissivity is the major source of uncertainty. Emissivity can be obtained from reflectivity (R), and, if the object is not transparent for the chosen wavelength, then ε = (1 – R). A Pyrolaser instrument (Pyrometer Instrument Co., Windsor, N.J.) measures emissivity at 865 nm in situ from the power of projected and reflected laser beams.10
Two-color pyrometry measures intensity of radiation on two spectral bands, assuming emissivity is identical at the chosen wavelengths. Temperature is derived from the ratio of intensities rather than their absolute values. However, any differences in effective emissivities between chosen wavelengths will have a large effect on accuracy of the measurements. Luckily, at temperatures greater than the reach of Pt-Rh thermocouples, emitted radiation can be registered in the visible range (Figure 3), for which sensors and optics represent well-developed technology.
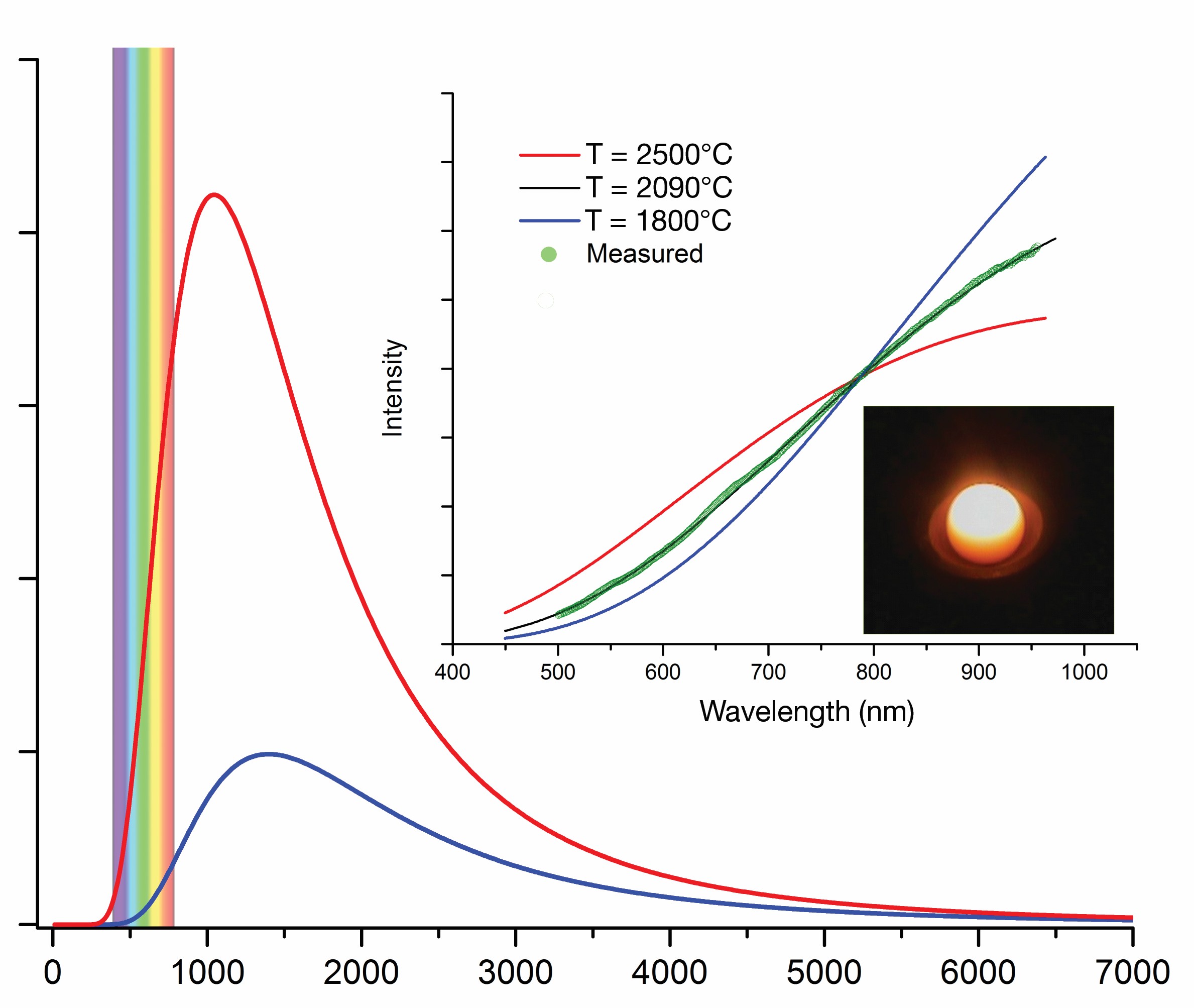
Figure 3. Theoretical spectra of blackbody radiation at 1,800°C and 2,500°C. Emissivity-independent fit of surface temperature of levitated bead from spectropyrometer is shown on the inset. Credit: Alexandra Navrotsky and Sergey V. Ushakov
Spectropyrometers, which use all the measured intensities for calculation of surface temperature, often are used in research and have been available commercially for more than a decade.11 When spectral responsivity and linearity corrections have been established through calibration, the programmed algorithms of data processing can be applied to derive a temperature value from the entire spectrum. The possibility to adapt conventional and high-speed complementary metal–oxide semiconductor cameras for 2-D and stereoscopic pyrometry was demonstrated recently.12 Although it is unlikely that radiation thermometry will ever reach accuracies of resistance and thermocouple contact measurements, the possibility of routine measurement of surface temperatures with uncertainties of ±5°C using spectropyrometry and in situ emissivity methods is a big advancement.
Thermal analysis and laser flash method
Methods of differential thermal analysis (DTA) and differential scanning calorimetry (DSC) take their root in measurement of sample temperature heating or cooling. Heat flow traces in DTA and DSC correspond to differences in temperature between a sample and an empty reference crucible. The methodology is well developed and widely used for biological materials, polymers, and ceramics up to 1,600°C with Pt-Rh thermocouples for temperature measurement. For the higher temperature range, DTA for operation to 2,400°C with W-Re thermocouples is available from several manufacturers. Nevertheless, the method generally was not used to quantify heat effects above 1,600°C because of fast aging of W-Re thermocouples affecting temperature calibration and the absence of standards for fusion enthalpy above the melting temperature of Al2O3 (2,054°C).
Recently, the Thermochemistry Laboratory at the University of California, Davis further explored the DTA method for its potential to provide the means to fill missing data on fusion and phase transition enthalpies above 1,600°C. A Setaram (Caluire, France) Setsys 2400 DTA equipped with graphite furnace and W-WRe sensor was modified to allow operation to 2,500°C and measurement of sample temperature by radiation thermometry (Figure 4). Enthalpies of phase transitions in HfO2 and La2O3 and fusion enthalpies of La2Zr2O7, LaAlO3, and La2O3 were measured successfully on samples sealed in tungsten crucibles to prevent reactions with carbon present in the furnace.7 The recent independent measurement of fusion enthalpy of Y2O3 allows high-temperature sensitivity calibration, which extends DTA capabilities to provide thermodynamic data above 2,000°C.13
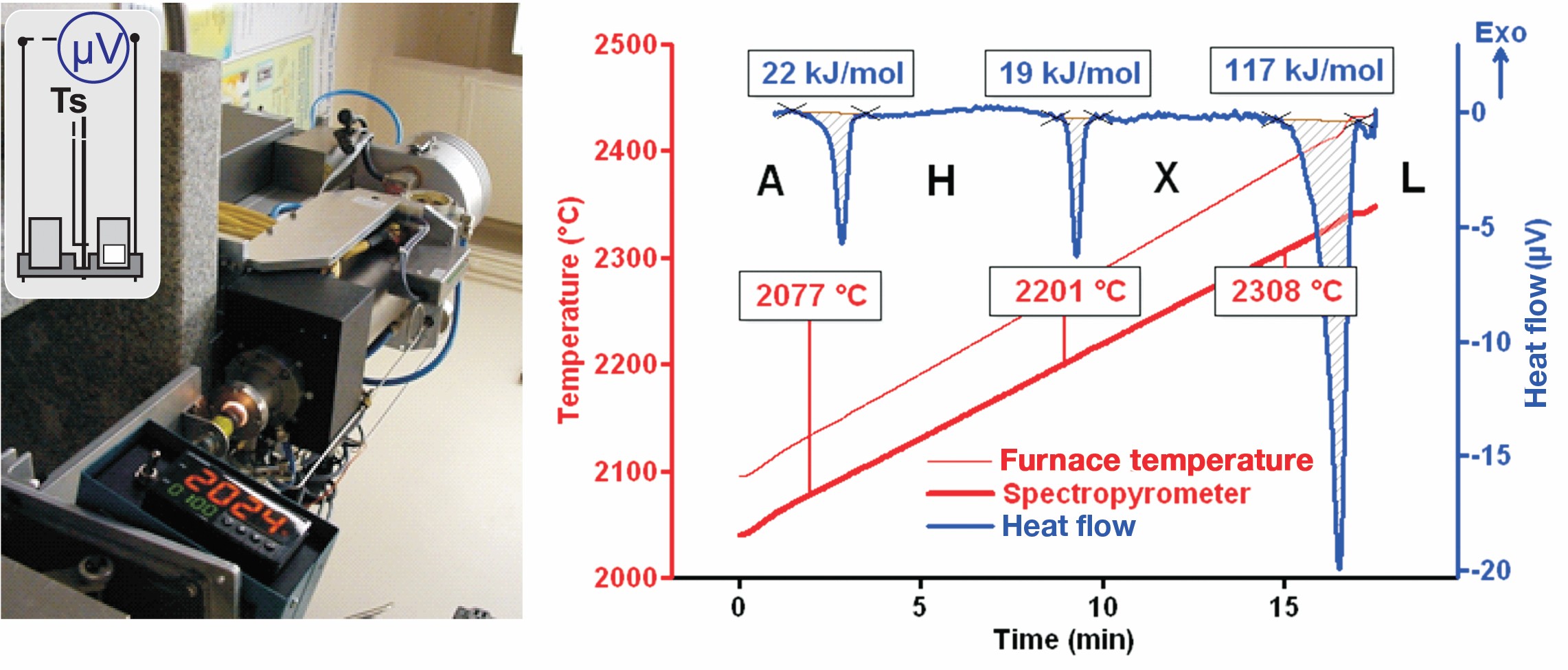
Figure 4. Modified high-temperature differential thermal analyzer (left) and heat flow trace (right) of Nd2O3 showing premelting phase transformations and melting (baseline subtracted). Credit: Alexandra Navrotsky and Sergey V. Ushakov
The laser flash method heats a disk-shaped sample by laser pulses on one side while tracing temperature rise on the opposite side. Many companies offer flash instruments for measurements up to 2,800°C. Originally the method was demonstrated to yield thermal diffusivity and heat capacity, which allows calculations of thermal conductivity.14 However, it is currently used almost exclusively for thermal diffusivity measurements. This is because heat capacity determination with laser flash requires a reference standard with the same surface emissivity as the sample or requires coating the sample and standard to provide identical emissivity, which is not required for thermal diffusivity measurements.
A common practice is to measure thermal diffusivity with a laser flash method and obtain heat capacity values from scanning calorimetry or estimate values from available reference data or theoretical considerations. Applications of the laser flash method for high-temperature heat-capacity measurements should be explored further using advances in radiation thermometry discussed above.
High-temperature calorimetry
Levitation calorimetry on metals using magnetic levitation and inductive heating has been practiced by several groups, and fusion enthalpies of platinum, palladium, molybdenum, neodymium, and 10 other metals were measured by 1975.6 These data, in addition to more recent independent measurements of heat contents by pulse heating,8 provide a reliable foundation for computational engineering of alloys.
Multiple compilations of thermodynamic data often refer to a single source when it comes to heat content of refractory oxides above 2,000°C.15 In this work, samples that weigh tens of grams were suspended in a graphite furnace and dropped into an ice-water calorimeter.15 Drop calorimetry also was used to measure heat content and fusion enthalpy on UO2 samples sealed in tungsten containers.16 However, no experimental measurements of fusion enthalpies of fusion of high-melting oxides, such as HfO2 and ZrO2, have been reported. The most recent assessments of thermodynamic functions for rare-earth oxides for calculations of phase diagrams is based on a single, sparingly described measurement of Y2O3 fusion enthalpy.13
A new technique for ultra-high-temperature drop calorimetry on ceramic samples was recently developed at UC, Davis (Figure 5). In this method, called drop-n-catch (DnC) calorimetry, samples are laser heated in a splittable nozzle aerodynamic levitator and dropped into a calorimeter at room temperature. The step in enthalpy versus temperature curve gives the heat of fusion.17 DnC was validated by measuring fusion enthalpy of Al2O3 and now is used in combination with DTA for enthalpy measurements of fusion and phase transitions of rare-earth oxides.
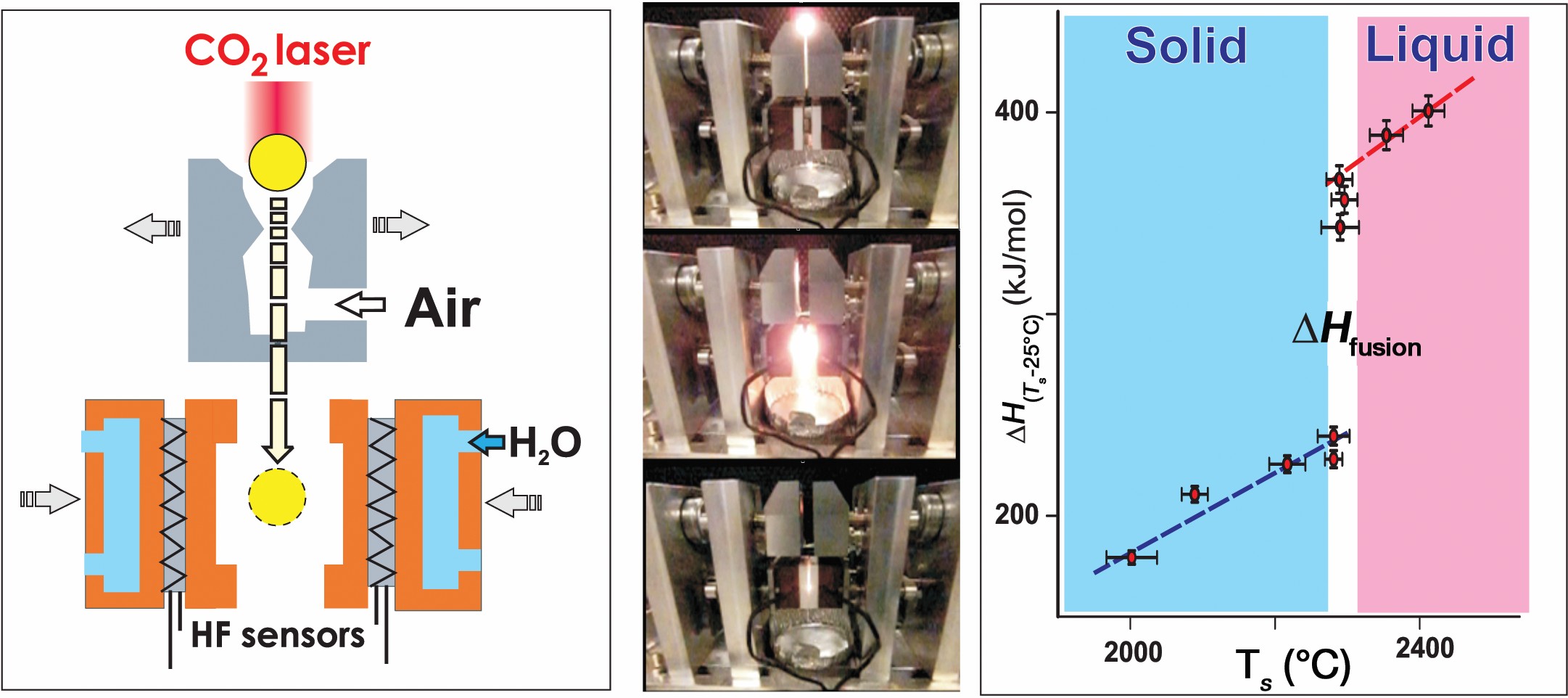
Figure 5. Drop-n-catch calorimeter schematic, operation and enthalpy versus temperature curve for Al2O3. Diameter of sample beads is 2.5–3 mm. Credit: Alexandra Navrotsky and Sergey V. Ushakov
Surface energies, perhaps an abstract concept for ceramic engineers, however, invariably come into play in models of mechanical properties and microstructure development. Thus, it is no surprise that surface and interfacial energies of metals and alloys are studied much more comprehensively than for ceramic materials. Thermodynamically, surface energy can be defined as a reversible work to create the new surface and interfacial energy as a sum of surface energies minus work of adhesion required to separate the interface. These figures are required as inputs for modeling of nucleation and fracture, in which new interfaces and surfaces are created—and of the sintering process in which they are eliminated.
High-temperature oxide melt solution calorimetry has been used for half a century for measurements of enthalpies of formation of refractory compounds, which are not easily dissolved in acids.9 In the past 20 years, it also became instrumental for measurements of surface energies of key refractory oxides: Al2O3, TiO2, ZrO2, HfO2, MgO, and Y2O3.9 The method is elegant in its simplicity—single-phase samples of various surface areas are dissolved in a sodium molybdate (3Na2O-4MoO3) or lead borate (2PbO-B2O3) melt at 700°C or 800°C. When the surface area is the only variable in the experiments, linear dependence of heat effects on surface area is observed and absolute value of the slope corresponds to surface energy.
Refractory oxides can manifest a strikingly large variation in surface energy between various polymorphs. For example, surface energy for ZrO2 changes from 0.5 J/m2 for the amorphous phase to above 3 J/m2 for the monoclinic phase. High-temperature phases of given oxides generally have lower surface energies, thus, amorphous and high-temperature polymorphs are thermodynamically stabilized at high surface areas. A familiar example is the stable amorphous Al2O3 layer that forms on the surface of aluminum-containing alloys (and is used as a starting layer for TBC engineering).
Surface energies are increasingly calculated from first principles. However, with ceramic materials representing more variation in bulk and surface structures and types of defects, experimental benchmarks are essential. Novel approaches to evaluate surface energy from heat evolved on coarsening in DSC and from enthalpy of water adsorption have been developed and validated against the values measured by solution calorimetry.18
X-ray and neutron diffraction
Powder X-ray diffractometry (XRD) has become an indispensable workhorse of materials characterization at all steps of ceramic development and manufacturing. In part, this is because of an accumulated collection of more than 170,000 entries of structural data for inorganic compounds and automated routines for whole profile Rietveld analysis, allowing quantification of phase contents and crystallite sizes. High-temperature powder XRD provides data on thermal expansion and volume change on phase transition, which are required for thermodynamic modeling. XRD also can be used for in situ phase diagram determination, especially at high temperatures, because of fast equilibration.
For powder XRD at temperatures lower than 1,700°C, Pt-Rh thermocouples and ceramic holders or Al2O3 capillaries19 at synchrotron sources can be used for measurements in variable atmospheres. For diffraction experiments at temperatures higher than 2,000°C, traditional measurements are limited to inert atmospheres using W-Re thermocouples and tungsten, molybdenum, or ruthenium sample holders or wires, which often also serve as heaters. Because of high thermal expansion of metal holders, the quality of diffraction data at temperatures in excess of 2,000°C is rarely suitable for structure determination.
High-temperature structures have been determined successfully from neutron diffractometry using a large volume of sample encapsulated in a container. Neutrons have the advantage over X-rays in that they are much more sensitive to light elements, including oxygen and hydrogen. However, because of scarcity of neutron sources around the world and complexity of measurements, such determinations have been limited until recently. For example, among rare-earth sesquioxides, high-temperature structures have been determined only for La2O3 and Nd2O3. However, this field is growing because much smaller sample sizes are now required for neutron diffractometry and facilities are becoming much more user-friendly.
Aerodynamic levitators with laser heating have been used for more than a decade at synchrotron and neutron sources worldwide to study structures of oxide melts.7,20 These operational instruments also can provide high-temperature diffraction data on solids. In levitation experiments, a sample is suspended in a gas flow and heated by a CO2 laser beam, and surface temperature is measured with radiation thermometry (Figure 6). Samples are usually prepared by laser melting of powders, in which case spheroids are formed by surface tension of the melt. Diffraction is obtained from the top of the bead levitated in a shallow conical nozzle. The technique can obtain high-energy synchrotron diffraction patterns in 0.1 s, while tens of minutes of collection time are required for neutron diffraction.4

Figure 6. Aerodynamic levitator at Argonne Photon Source (beamline 6-ID-D) for in situ high-temperature diffraction on ceramic materials. Matthew Fyhrie (UC Davis) is loading the oxide sphere in the levitator chamber with an air pick. Credit: Alexandra Navrotsky and Sergey V. Ushakov
Because the levitating sample is heated from the top and cooled from the bottom by gas flow, thermal gradients in the diffracted volume are unavoidable. They are larger for solid samples than for melts because of the absence of thermal conduction inside the sample by convective flows, and can reach 150°C. Despite such problems, the first applications of the method resolved some lingering questions on structure and thermal expansion of refractory oxides, such as high-temperature phase transformations in Y2O3 and Yb2O3, and thermal expansion of YSZ and La2Zr2O7 to melting temperatures.4,7 The method now is used to measure thermal expansion and volume changes on phase transformation in rare-earth oxides, which are critical for CalPhaD modeling of oxide systems with rare earths.
The unique advantages of levitation are elimination of sample reaction with container or support material and flexibility in choice of atmosphere. UHTC formulations for flight-related applications should be capable to sustain high-temperature exposure to oxygen in conditions of hypersonic flow. In-depth understanding of high-temperature interaction with water vapor is critical for propulsion-related UHTC formulations and for design of protective coatings for nuclear applications. Diffraction on levitated, laser-heated samples in variable environments at synchrotron and neutron user facilities provides versatile new tools for high-temperature research. It can provide high-temperature thermophysical data (thermal expansion and volume change on phase transitions) and, potentially, kinetic data on high-temperature oxidation reactions and transformations.
Most high-temperature thermodynamic and thermophysical data on refractory oxides were obtained during the Cold War era using ad hoc instrumentation in specialized laboratories. The current availability of synchrotron and neutron user facilities, turnkey lasers, high-temperature DTA, and commercial calorimeters now make high-temperature research sustainable in university settings. When required basic thermodynamic inputs are available, computational materials design has potential to speed up the “lab-to-fab” cycle for materials development. It is our hope that such developments will be fruitful, and our current students will witness civilian supersonic flights, manned landings on Mars and other regions of the solar system, and improvements in technology for sustainability on Earth that older generations could only dream of.
Think big, bold, and hot!
Acknowledgements
High-temperature experiments on rare-earth oxides at UC Davis were supported by National Science Foundation Award DMR-1506229. Use of the Advanced Photon Source (APS, beamline 6-ID-D), a DOE Office of Science user facility operated by Argonne National Laboratory, was supported by DOE Contract No. DE-ACO2-06CH11357
Capsule summary
An opportunity for ceramic materials
Although measurement and modeling techniques have accelerated development of superalloys, these materials are reaching their high-temperature limits. Ceramic materials offer unprecedented higher-temperature capabilities, but lack experimentally measured thermodynamic property data.
Inside this article
We review recent developments in experimental methods that can provide data for high-temperature properties of refractory ceramics, including thermal analysis and the laser flash method; high-temperature calorimetry; and X-ray and neutron diffraction.
Big, bold, and hot
Recent advances have made high-temperature property research more available and sustainable than ever before. Using newly acquired thermodynamic data, computational design has an incredible potential to develop new ceramic materials.
Cite this article
A. Navrotsky and S. V. Ushakov, “Hot matters—Experimental methods for high-temperature property measurement,” Am. Ceram. Soc. Bull. 2017, 96(2): 22–28.
About the Author(s)
Alexandra Navrotsky is director of the Peter A. Rock Thermochemistry Laboratory and NEAT ORU at the University of California, Davis. Sergey V. Ushakov is staff research associate in the Peter A. Rock Thermochemistry Laboratory at UC, Davis. Contact Navrotsky at Anavrotsky@ucdavis.edu.
Issue
Category
- Engineering ceramics
- Refractory ceramics
Article References
1B. Sundman, U.R. Kattner, C. Sigli, M. Stratmann, R. Le Tellier, M. Palumbo, and S.G. Fries, “The Open Calphad thermodynamic software interface,” Comput. Mater. Sci., 125, 188–96 (2016).
2M. F. Horstemeyer, Integrated computational materials engineering (ICME) for metals: Using multiscale modeling to invigorate engineering design with science, Wiley, New York, 2012.
3N.P. Padture, “Advanced structural ceramics in aerospace propulsion,” Nat. Mater., 15 [8] 804–809 (2016).
4S.V. Ushakov, A. Navrotsky, R.J.K. Weber, and J.C. Neuefeind, “Structure and thermal expansion of YSZ and La2Zr2O7 above 1500°C from neutron diffraction on levitated samples,” J. Am. Ceram. Soc., 98 [10] 3381–88 (2015).
5T. Huepf, C. Cagran, and G. Pottlacher, “Thermophysical properties of 22 pure metals in the solid and liquid state—The pulse-heating data collection,” EPJ Web Conf., 15 01018 (2011).
6M.G. Frohberg, “Thirty years of levitation melting calorimetry—A balance,” Thermochim. Acta, 337 [1–2] 7–17 (1999).
7S.V. Ushakov and A. Navrotsky, “Experimental approaches to the thermodynamics of ceramics above 1500°C,” J. Am. Ceram. Soc., 95, 1463–82 (2012).
8W. Schroeer and G. Pottlacher, “Estimation of critical data and phase diagrams of pure molten metals,” High Temp.-High Pressures, 43 [2–3] 201–15 (2014).
9A. Navrotsky, “Progress and new directions in calorimetry: A 2014 perspective,” J. Am. Ceram. Soc., 97 [11] 3349–59 (2014).
10E.S. Cameron, “Laser assisted remote temperature measurement”; pp. 155–62 in Optomechanical design of laser transmitters and receivers, SPIE Proceedings, Vol. 1044, 1989.
11R.A. Felice, “The spectropyrometer—A practical multi-wavelength pyrometer,” AIP Conference Proceedings, 684 [Pt. 2] 711–16 (2003).
12M. Jakob, P. Lehnen, P. Adomeit, and S. Pischinger, “Development and application of a stereoscopic 3-color high-speed ratio-pyrometry,” Combust. Flame, 161 [11] 2825–41 (2014).
13D. Kapush, S. Ushakov, A. Navrotsky, Q.-J. Hong, H. Liu, and A. van de Walle, “A combined experimental and theoretical study of enthalpy of phase transition and fusion of yttria above 2000°C using ‘drop-n-catch’ calorimetry and first-principles calculation,“ Acta Mater., 124, 204–209 (2017).
14W.J. Parker, R.J. Jenkins, C.P. Butler, and G.L. Abbott, “Flash method of determining thermal diffusivity, heat capacity, and thermal conductivity,” J. Appl. Phys., 32 [9] 1679–84 (1961).
15C D. Pears, “The thermal properties of twenty-six solid materials to 5000 DegF. or their destruction temperatures”; pp. 420 pp. in Technical Documentary Report, Vol. ASD-TDR-62-765. Southern Research Institute, Birmingham, Alabama, 1963.
16R.A. Hein, P.N. Flagella, and J.B. Conway, “High-temperature enthalpy and heat of fusion of uranium dioxide,” J. Am. Ceram. Soc., 51, 291–92 (1968).
17S.V. Ushakov, A. Shvarev, T. Alexeev, D. Kapush, and A. Navrotsky, “Drop—and—catch (DnC) calorimetry using aerodynamic levitation and laser heating,” J. Am. Ceram. Soc. (approved for publication Oct. 24, 2016), in press.).
18S. Hayun, T. Roehling, S.V. Ushakov, and R.H.R. Castro, “Experimental methodologies for assessing the surface energy of highly hygroscopic materials: The case of nanocrystalline magnesia,” J. Phys. Chem. C, 115, 23929–35 (2011).
19R.P. Haggerty, P. Sarin, Z. D. Apostolov, P.E. Driemeyer, and W.M. Kriven, “Thermal expansion of HfO2 and ZrO2,” J. Am. Ceram. Soc., 97 [7] 2213–22 (2014).
20J.K.R. Weber, C.J. Benmore, L.B. Skinner, and J.B. Parise, “Measurements of liquid and glass structures using aerodynamic levitation and in-situ high-energy X-ray and neutron scattering,” J. Non-Cryst. Solids, 383, 49–51 (2014).
Related Articles
Market Insights
Engineered ceramics support the past, present, and future of aerospace ambitions
Engineered ceramics play key roles in aerospace applications, from structural components to protective coatings that can withstand the high-temperature, reactive environments. Perhaps the earliest success of ceramics in aerospace applications was the use of yttria-stabilized zirconia (YSZ) as thermal barrier coatings (TBCs) on nickel-based superalloys for turbine engine applications. These…
Market Insights
Aerospace ceramics: Global markets to 2029
The global market for aerospace ceramics was valued at $5.3 billion in 2023 and is expected to grow at a compound annual growth rate (CAGR) of 8.0% to reach $8.2 billion by the end of 2029. According to the International Energy Agency, the aviation industry was responsible for 2.5% of…
Market Insights
Innovations in access and technology secure clean water around the world
Food, water, and shelter—the basic necessities of life—are scarce for millions of people around the world. Yet even when these resources are technically obtainable, they may not be available in a format that supports healthy living. Approximately 115 million people worldwide depend on untreated surface water for their daily needs,…




