Zirconium is a crucial component of refractories used in the glass industry because it enables the design of materials with high strength at elevated temperatures, low thermal conductivity, high resistance against thermal shock, and minimal reaction with molten glass.
Despite their good behavior, zirconium-based refractories still must be replaced periodically due to wear, primarily because of a complex corrosion phenomenon that combines three general physical and chemical processes: penetration, dissolution, and erosion.1
In this paper, the corrosion of zirconium-based refractories used as a glass contact material in melting tanks is described and discussed.
Basics of zirconium-based refractories
General classification
Zirconium-based refractories are generally classified into three types:
- Dense zircon refractories are characterized by a typical composition of alumina (0.0–2.5 wt.%), zirconia (64–68 wt.%), silica (30–35 wt.%), and minor concentrations of other components such as Fe2O3 and Na2O. Their microstructure is simple and consists mainly of sintered zircon (ZrSiO4) particles with small pores.
- Fused cast alumina–zirconia–silica (AZS) refractories are composed of around 43–50 wt.% alumina, 33–42 wt.% zirconia, and 13–20 wt.% silica, with the possibility of having small amounts of other components such as B2O3, Na2O, Fe2O3 or TiO2. Their microstructure consists of zirconia grains and domains of alumina-zirconia eutectic composition embedded in a glassy aluminosilicate phase.
- Fused cast high zirconia (HZ) refractories, similar to dense zircon refractories, are used when the quality and corrosion requirements are especially demanding. These materials are characterized by a composition rich in zirconia (above 94 wt.%) as their main component and other components are 1.2 wt.% of alumina and 4 wt.% of silica, indicating a small vitreous phase content (about 6%). Unlike AZS refractories, the microstructure is formed by zirconia grains and a low content of glassy phase, which is identified as a thin interface layer along grains.2
Thermomechanical properties
During service, refractory materials are subjected to large temperature changes up to 1,600°C. Throughout the heating process, the material’s microstructure undergoes changes that can slightly modify its behavior compared to the material at ambient conditions. In zirconium-based refractories, one of the most important changes during heating up is the phase transformation that zirconia (ZrO2) undergoes, typically at a temperature near 1,157°C. During this transformation, its structure changes from monoclinic to tetragonal, resulting in a decrease in volume without any change in its chemical composition.
This transformation is one of the major issues in zirconia-based refractories because this change is responsible for microcrack generation on the material microstructure, which can lead to future failure. This phenomenon particularly affects the AZS and HZ refractories, as zirconia is a principal component of their microstructure. To mitigate volume-change effects and reduce crack formation, the glassy phase is incorporated into these materials.
Generally, thermal shock and zirconia transformation may result in a decrease in the mechanical properties of the refractories, mainly due to the generation and extension of cracks on their microstructure. However, the corrosion resistance is also impacted because these defects result in free spaces that can be filled by molten glass, penetrating the refractory easily and accelerating its wear. Hence, a refractory that can keep a dense microstructure as free as possible of pores and cracks, even after temperature variations, may exhibit better resistance against glass penetration and increase its corrosion resistance.
Manufacturing techniques
The microstructure of refractories is influenced by manufacturing parameters, which in turn significantly affect their properties. As a result, the manufacturing process plays a crucial role in determining the overall performance of the refractory.
In general, two primary methods are used to manufacture zirconium-based refractories: the fused cast process and sintering. The first one is used predominantly for the manufacture of AZS and HZ refractories. This process starts with mixing raw materials that are melted in an arc furnace and then poured into molds to anneal and solidify. The blocks are later removed from the molds, inspected, and finished.3 This process, in general, allows manufacturers to obtain higher densities and lower apparent porosity (0–2%) in the refractories, but it may be more expensive than other refractory manufacturing processes.
On the other hand, the sintering process is commonly used to manufacture dense zircon refractories. This process involves the consolidation of the raw material (powder) through forming and sintering. Although this process may exhibit higher apparent porosity (10–19%) than the fused cast method, when mechanical pressing is applied, the use of fine raw materials and more advanced processes such as hot isostatic pressing can reduce porosity. Likewise, it offers more flexibility in composition and can be used for particular applications.
Corrosion in zirconium-based refractories
Because of their different microstructures, phases, and components, the corrosion mechanisms for zirconium-based refractories may change from one type to another.
Corrosion in AZS refractories occurs at high temperatures when alkali elements, such as sodium, present in the molten glass start to diffuse into the glassy phase, causing its exudation and pore formation. This exudation also leads to both the dissolution of alumina in the corundum phase and the release of less soluble zirconia crystals, a process that occurs in a boundary layer formed as a result of the reaction between the refractory and molten glass (Figure 1). The generation of pores causes AZS refractories to lose their permeability, and molten glass can diffuse into the refractory, increasing the corrosion rate progressively until the refractory material fails.
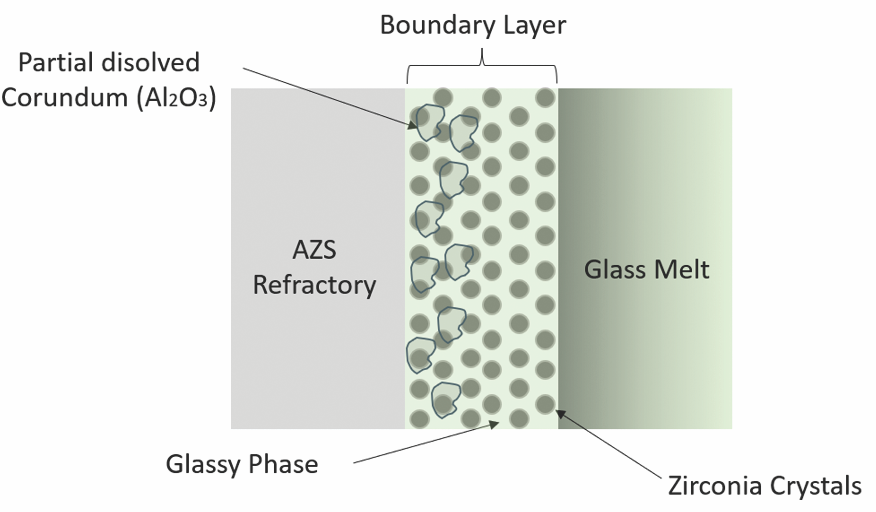
Figure 1. Illustration of the interface between AZS refractory and glass melt, showing the boundary layer where partial dissolution of corundum-phase alumina occurs and zirconia crystals migrate into the glass melt. Credit: Perez Velasquez et al., ACT (CC BY-NC 4.0)
In contrast, the corrosion in dense zircon refractories is governed by the decomposition of zircon crystals at high temperatures into zirconia and silica, which may be dissolved and combined with glass components.4 Generally, as a result of the high solubility of silica in the glass, it rapidly dissolves in the interface, while the zirconia crystals with a lower solubility remain surrounded by glass. Hence, the glass surrounding the zirconia is characterized by a different chemical composition from the initial glass (resulting from its mixing with silica and zirconia), which alter some of its properties.
Corrosion processes can also vary depending on the kind of glass that is in contact with the refractory as well as thermal conditions, among other factors. Guzman et al. investigated wear on AZS bricks located in a glass tank after several years of service in contact with soda lime silicate glass.5 They summarized the corrosion process in four well-defined stages: penetration of alkali elements, exudation of glassy phase, cracking, and erosion. The alkalis from the glass penetrate the alumina–zirconia and zirconia phases, reacting with alumina to form a sodium aluminosilicate layer. These layers, which are characterized by high surface tension, enhances exudation of the glassy phase and ultimately lead to the formation of cavities and cracks that reduce the strength of the refractory.
In addition, the corrosion process in special glasses (e.g., borosilicate, aluminosilicate, lead silicate, phosphate glasses) has also been studied. Hong Li et al. studied the corrosion of AZS refractories by calcium aluminosilicate and soda-lime silicate glasses,6 which are utilized in the development of glass-ceramics at temperatures up to 1,480°C under static conditions. In general, the viscosity of these glasses decreased rapidly at temperatures above 1,200°C and then could penetrate the AZS microstructure. In the beginning, calcium oxide and sodium oxide are diffused into the refractory, and liquidus temperature decreases at the glass–refractory interface, forming new mineral phases such as anorthite and gehlenite, which later are washed out, causing continuous corrosion of the refractory.
Corrosion tests: Current approaches and limitations
Refractory corrosion tests are mainly classified into two groups: static tests, in which the motion of fluid in contact with refractory is not simulated, and dynamic tests, in which the refractory and fluid are in relative motion. In both cases, simulating the exact conditions of service life is difficult due to the overwhelming number of variables to consider, including sample geometry and size, stress distribution, time and thermal gradient, among other variables. However, testing small scales is cheaper than field trial testing, which provides impetus for researches to improve on these techniques.
Examples of new or improved corrosion testing include the continuous wear testing device developed by Kircher et al.7 This technique, based on the rotating cylinder test, evaluates the corrosion under dynamic conditions by analyzing different parameters simultaneously during the test, such as the weight of the samples as a function of time and sample surface. Also, Poirier et al. proposed the use of X-ray diffraction at high temperatures combined with Rietveld quantification to determine the corrosion kinetics.8 This combination allows evaluation of the mechanisms involved and the phase changes in the process, which could help improve the knowledge and prediction of the phenomena of refractory degradation.
Even with these improvements, the most significant modeling efforts to refractories by various authors pertain to studies performed in the steel and metals industry. This focus highlights a clear lack in models that can elucidate the corrosion models and tests for glass industries. To enable the development of better refractory corrosion models and tests for glass manufacturers, we must improve our understanding of the thermodynamic and kinetic processes involved in the corrosion of zirconium-based refractories by molten glass.
Considerations for understanding the corrosion of zirconium-based refractories
Thermal stability and temperature effects
In principle, the melting temperatures of refractories are a good starting point for corrosion evaluation, as they strongly influence phenomena such as diffusion in the host material. Therefore, at least initially, compositions with higher melting points may have a longer service life, so thermal stability plays a crucial role in understanding refractory corrosion resistance.
In the case of zirconium-based refractories, it is well known that high temperatures may result in the dissociation of zircon crystal into silica and zirconia. Although there is an essential variation in the decomposition temperature across different studies (varying from 1,540–1,683°C), all studies have concluded that the reaction accelerates dramatically with rising temperature. The substantial variations observed may be attributed to other factors such as refractory composition, purity, the refractory density, the stabilizing oxide, and particle size. It has been demonstrated that even small amounts of impurities such as iron, titanium, and alkalis increase the degradation of zircon, and decreasing particle size can lead to dissociation reactions occurring at lower temperatures than expected.9,10
The dissociation of zircon into silica and zirconia may not only increase molten glass penetration within the refractory, as described earlier in the section on corrosion in zirconium-based refractories, but may also result in a decrease in the electrical resistivity of the refractory surface. In electrical furnaces, this decrease may produce changes within the current path because the current can now be conducted through the refractory block’s surface instead of molten glass (Figure 2). Such a change in the current path can accelerate wear, which can lead to glass leakage and process issues due to nonhomogeneous heating in the tank.
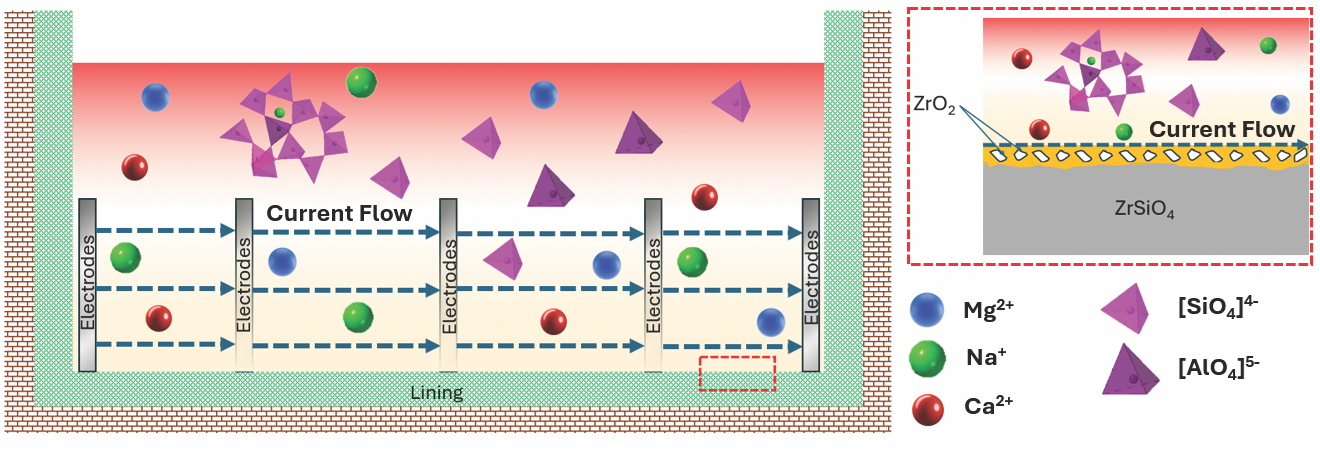
Figure 2. Interaction between molten glass and the furnace lining in electric furnaces, illustrating the change in current flow path caused by zircon decomposition. The inset on the right highlights the formation of a zirconia layer at the refractory surface and the migration of ionic species (Na+, Ca2+, Mg2+) and structural units ([SiO4]4-,[AlO4]5-), which accelerate wear. Credit: Perez Velasquez, Montazerian, and Mauro
Thermodynamics
The thermodynamic approach is an essential tool in the study of refractory corrosion because it allows researchers to elucidate different important parts of the process, such as the reactions that can take place, the products that can be formed, and the solubility of different components, among others.
Although a nonnegligible number of thermodynamic studies and approaches have been applied to the glass industry, most of them have been focused on the corrosion of refractories by vapors and gases used on the superstructure and crown of the furnaces13,14; the application of thermodynamic calculations to study the corrosion on contact glass refractories, such as the zirconium-based materials, is limited. Therefore, there is a notable opportunity to increase the application of these methodologies to analyze the corrosion potential and processes in different glass systems.
We recently studied the effect of [CaO]+[Al2O3] content within a group of fiberglass compositions on the zircon refractory decomposition. The results indicated that, although there is no linear trend, in general, the increasing of [CaO]+[Al2O3] results in a lower zircon breakdown temperature, with drops up to 350°C compared with the typical value for pure zircon (1,673±10°C). Furthermore, thermodynamic calculations revealed that Na2O is the most reactive oxide when interacting with zircon, obeying the acidobasicity relationship between the refractory and the glass oxides, so that more basic components (e.g., sodium oxide, calcium oxide, and magnesium oxide)
can react more strongly than the less basic and acidic glass oxides (e.g., alumina and titania) with zircon, characterized by acidic nature.
Kinetics
Zirconium-based refractories are soluble in the melts they contain, so they will eventually dissolve entirely. Thus, the dissolution rate is an essential parameter to define when the corrosion process is evaluated.
Considering that the dissolution reaction involves diffusion and can also be affected by the viscosity of the molten glass, the temperature dependence of the process is evident. Hence, this dependence is usually represented by the Arrhenius equation. However, not all systems can be fit by an Arrhenius plot because they exhibit behavior far from exponential temperature dependence, as is the case for mullite.15 Therefore, the Arrhenius dependence should be used only in specific cases when the liquid is far from being saturated with components from the solid.
During glass making, the refractories that are in direct contact with the glass (e.g., bottom, sidewall, throat) can experience different types of forces generated by density and surface tension variations within the melt, as well as by mechanical forces generated by equipment in the tank. These forces have an evident impact on the dissolution reaction, and so the dissolution of the refractories commonly considers four possible cases16: stagnant liquid or molecular diffusion, natural or free convection, forced convection, and surface tension gradient driven.
The corrosion on refractories is commonly assumed to be a mass-transport governed process, where various temperature-dependent variables, such as viscosity, density, diffusion coefficients, and concentration gradients, may influence the dissolution rates of the refractory components. In addition, the theoretical dissolution rates for different conditions that can take place within the melting furnaces have been established and validated experimentally for ceramic materials by a few authors.17–19
However, many of the refractories used in the glass industry contain three or more phases, and many of the glass compositions are multicomponent systems. So, obtaining the values of some of the parameters experimentally is complicated. Therefore, most times theoretical models cannot accurately estimate the dissolution rates. Hence, to extend and enhance the kinetics approach, it is suggested to look for experimental methods that allow parameters to be acquired easily, develop new models or update existing ones, and accompany this kind of analysis with thermodynamics and thermal stability considerations.
Effect of glass composition
The chemical composition of the glass is another critical parameter to consider when analyzing the corrosion process in zirconium-based refractories. Generally, we can assume that components with a similar nature are less prone to react to each other, while on the contrary, important differences likely result in a chemical reaction; hence, from that previous statement, an initial classification in acid and basic oxides can be considered.
With dense zircon refractories, thermal decomposition dominates the corrosion process, where the breakdown temperature may vary depending on the components of the molten glass. Mauro et al. mention that, as a general rule, glass compositions with high reactivity with zircon will show lower breakdown temperatures than the more inert glass compositions.20
From a reaction viewpoint, the decomposition of zircon into zirconia and silica provides an opportunity for those dissolution products to combine with other components in the glass melt.
Kato and Araki investigated the effect of glass composition on the corrosion of zirconia and zircon refractories.4 They found that alkali components, such as sodium oxide, are the most corrosive for both refractory types. Furthermore, in the case of zirconia, the corrosion rate increased significantly due to the presence of CaO.
In the case of the AZS refractories, the main chemical interaction between the refractories and the melt is the action of the alkali-earth and alkali elements in glass. According to Guloyan and Pustyl’nikov,21 these ions initially penetrate the refractory through the glassy phase, decreasing its viscosity and generating a breakdown of structure cohesiveness. Similarly, new components, such as alkali aluminosilicates, can be formed through the interaction of alkalis, primarily sodium ions, with refractory components (e.g., alumina, zirconia, and silica). Furthermore, ions such as Al3+, Zr4+, and O2– also appear due to the interaction, influencing the thickness and density of the boundary layer in the interaction zone.
Min’ko et al. found after static corrosion tests between 1,440–1,500°C that the most aggressive components for zirconium-based refractories are the oxides of boron, sodium, calcium, lead and barium.22 Moreover, the alkaline earth oxides tend to be more corrosive than alkaline oxides; hence, when high calcium glass compositions (CaO > 20 wt.%) are used, the zirconium-based refractories are insufficiently stable.
Due to the high number of glass compositions used in the glass industry, as well as the nonnegligible effects of even small changes on the composition in the corrosion rate of the refractories, it is not an easy task to achieve a complete and totally clear understanding of the impact of the glass composition on the corrosion process. However, based on previous studies, it can be discerned that alkali oxides such as sodium and potassium oxides, together with oxides such as calcium, sulfur, chromium, nickel, lead, barium, and iron, compose the group of the most corrosive agents to zirconium-based refractories, mainly because of their high mobility, basic nature and decreasing effect on the glass melt viscosity.
The future of zirconium-based refractory corrosion assessment
Zirconium-based refractories represent one of the best materials used as glass-contact refractories in melting furnaces. However, corrosion remains one of the most meaningful challenges, so it is critical to understand this process properly to increase the corrosion resistance of melting furnaces and precisely predict service life.
Nowadays, there is a considerable amount of knowledge regarding the corrosion process of zirconium-based refractories when employed in glass-contact areas, but questions about the process still remain. The development direction for zirconium-based refractory corrosion tests should be mainly focused on the following points:
- More studies on dense zircon refractories: Existing literature and research on zirconium-based refractories focuses on the fused cast refractories, AZS and HZ. There is thus an apparent gap regarding the dense zircon refractories, which are often used in industrial furnaces. Future research should be conducted to further understand dense zircon refractories and clarify the differences and similarities with respect to the other families. In addition, although there are general kinetics models and thermodynamics approaches to analyze the refractory corrosion, the development of new and specific models for zirconium-based refractories could be a valuable tool.
- Consideration for novel glass compositions: In the coming years, new glass compositions such as the aluminosilicophosphate (LionGlassTM® composition developed at The Pennsylvania State University) will enter commercial production to help meet global demands to reduce carbon impact. Therefore, the impact of different common oxides used in glass manufacturing on the corrosion of commonly used zircon-based refractories must be fully clarified so that the implementation of new compositions can be done without important process restrictions.
- Implementation of advanced computation tools: Recently, the use of computational tools has been increasing to evaluate refractory corrosion in other industries, such as in steel manufacturing. Nevertheless, the glass industry does not seem to follow the same trend, and limited research in this field has been done. To speed up the process and reach a superior understanding of the corrosion process in zirconium-based refractories, the implementation of computational tools such as machine learning, molecular dynamics, and Monte Carlo methods, which have been used to study other kinds of refractories in specific applications,23,24 should be encouraged.
- Exploring dopant benefits: Solubility of refractory components in molten glass plays one of the most important roles in corrosion of the refractories. Therefore, it is necessary to look for the inclusion of other known oxides with lesser solubility in the molten glass than zirconia, alumina, and silica, which represent the main components in the zirconium family of refractories. However, adding new components must ensure the main advantages of zirconium-based refractories in the manufacture of glass products, such as the minimization of defects and their versatility, to demonstrate a high performance against different types of glasses.
This article is excerpted and modified from “Corrosion of zirconium-based refractories in glass-contact areas: Mechanisms and challenges,” ACT 2025, 22(3): e15064.
Cite this article
C. P. Velasquez, M. Montazerian, and J. C. Mauro, “Corrosion of zirconium-based refractories by molten glass,” Am. Ceram. Soc. Bull. 2026, 105(2): 30–35.
About the Author(s)
Cristian Perez Velasquez is a Ph.D. candidate, Maziar Montazerian is assistant research professor, and John C. Mauro is the Dorothy Pate Enright Professor in the Department of Materials Science and Engineering at The Pennsylvania State University. Contact Mauro at jcm426@psu.edu.
Issue
Category
- Glass and optical materials
- Refractory ceramics
Article References
1J. Sune, B. Voicu, and J. Pär, “Corrosion mechanism and kinetic behaviour of refractory materials in contact with CaO–Al2O3–MgO–SiO2 slags,” Scandinavian Journal of Metallurgy 2005, 34(5): 283–292.
2G. Duvierre and Y. Boussant-Roux, “Fused zirconia or fused AZS: Which is the best choice?” 59th Conference on Glass Problems: Ceramic Engineering and Science Proceedings 2008, pp. 65–80.
3P. Sengupta, Refractories for the Chemical Industries. Springer International Publishing, 2020.
4K. Kato and N. Araki, “The corrosion of zircon and zirconia refractories by molten glass,” J. Non. Cryst. Solids 1986, 80(1–3): 681–687.
5A. M. Guzmán, D. I. Martínez, and R. González, “Corrosion–erosion wear of refractory bricks in glass furnaces,” Eng. Fail. Anal. 2014, 46: 188–195.
6H. Li, J. Cheng, and L. Tang, “Corrosion of electrocast AZS refractories by CAS glass-ceramics melting,” J. Non. Cryst. Solids 2008, 354(12–13): 1418–1423.
7V. Kircher, Burhanuddin, and H. Harmuth, “Design, operation and evaluation of an improved refractory wear testing technique,” Measurement 2021, 178: 109429.
8J. Poirier et al., “New advances in the laboratory characterization of refractories: Testing and modelling,” Metall. Res. Technol. 2017, 114(6): 610.
9R. S. Pavlik, H. J. Holland, and E. A. Payzant, “Thermal decomposition of zircon refractories,” J. Am. Ceram. Soc. 2001, 84(12): 2930–2936.
10Y. Kanno, “Thermodynamic and crystallographic discussion of the formation and dissociation of zircon,” Journal of Materials Science 1989, 24: 2415–2420.
11L. J. Manfredo and R. N. McNally, “Solubility of refractory oxides in soda‐lime glass,” J. Am. Ceram. Soc. 1984, 67(8): C‐155-C‐158.
12P. M. Toperesu et al., “Static and dynamic corrosion behavior of SnO2-doped AZS slip cast refractory in SLS glass,” Int. J. Appl. Ceram. Technol. 2022, 19(2): 1160–1170.
13R. H. Nilson et al., “Analytical models for high-temperature corrosion of silica refractories in glass-melting furnaces,” Glass Sci. Technol. 2003, 76: 136–151.
14R. G. C. Beerkens and O. S. Verheijen, “Reactions of alkali vapours with silica-based refractory in glass furnaces, thermodynamics and mass transfer,” Physics and Chemistry of Glasses 2005, 46(6): 583–594.
15B. N. Samaddar, W. D. Kingery, and A. R. Cooper, “Dissolution in ceramic systems: II, dissolution of alumina, mullite, anorthite, and silica in a calcium–aluminum–silicate slag,” J. Am. Ceram. Soc. 1964, 47(5): 249–254.
16G. A. Pecoraro, “How the properties of glass melts influence the dissolution of refractory materials,” in Properties of Glass-Forming Melts. CRC Press, 2005. p. 339.
17L. R. Schlotzhauer and J. R. I. Hutchins, “Corrosion resistance of alumina zirconia silica refractories,” The Glass Industry 1966, 47(1): 26–29.
18M. N. Kucheryavyi et al., “Resistance of refractory materials to vertical honeycomb corrosion,” Science for the Glass Industry (Translated) 1983, 40: 335–339.
19K. Schwerdtfeger, “Dissolution of solid oxides in oxide melts. The rate of dissolution of solid silica in Na2O-SiO2 and K2O-SiO2 melts,” J. Phys. Chem. 1966, 70(7): 2131–2137.
20J. C. Mauro et al., “Accelerating the design of functional glasses through modeling,” Chemistry of Materials 2016, 28(12): 4267–4277.
21Y. A. Guloyan and O. M. Pustyl’nikov, “Physical–chemical interactions during contact between molten glass and refractories,” Science for Glass Production (Translated) 2011, 67: 297–303.
22N. I. Min’ko and V. M. Nartsev, “Effect of the glass composition on corrosion of zirconium-containing refractories in a glass-melting furnace (a review),” Refractories for the Glass Industry (Translated) 2007, 64: 335–342.
23N. J. Smith-Gray et al., “Machine learning to predict refractory corrosion during nuclear waste vitrification,” MRS Advances 2021, 6: 131–137.
24J. D. Vienna et al., “Glass property–composition models for support of Hanford WTP LAW facility operation,” Pacific Northwest National Laboratory, July 2021.
*All references verified as of Jan. 23, 2026.
Related Articles
Bulletin Features
The nonferrous metals market: Supply and regulatory pressures inspire strategies for a resilient future
Nonferrous metals serve foundational roles in the electrification, renewable energy, and digital transformation. Nonferrous metals are metals that do not contain iron in significant amounts. These metals typically are nonmagnetic, corrosion resistant, electrically and thermally conductive, and lightweight, making them ideal for applications in the emerging markets mentioned above. Even…
Market Insights
Industrial digitalization: ‘Smart’ operations can improve worker safety and well-being in high-temperature environments
Heavy industry is the backbone of economies around the world, critical to automotive production, construction, the energy sector, and everything in between. But many heavy industries are facing worker shortages. There are more than 400,000 open manufacturing jobs in the United States, according to the Bureau of Labor Statistics.1 With…
Market Insights
‘Fail fast’ manufacturing: How disciplined experimentation strengthens, not threatens, quality
In manufacturing, few phrases raise eyebrows faster than “fail fast.” In the startup world, this business strategy is celebrated as a sign of agility. On a ceramic manufacturing floor, it can sound careless or even dangerous. In manufacturing, few phrases raise eyebrows faster than “fail fast.” In the startup world,…





