How does glass alter with time? For the last hundred years this has been an important question to the fields of object conservation and archeology to ensure preservation of glass artifacts.1 This same question is part of the development and assessment of durable glass waste forms for the immobilization of nuclear wastes. Researchers have designed experiments ranging from simple to highly sophisticated to answer this question and, as a result, have gained significant insight into the mechanisms that drive glass alteration. However, gathered data have been predominately applicable to only short-term alteration—i.e., over the course of decades.
Long-term mechanisms of glass alteration have remained elusive.2 These mechanisms are of particular interest to the international nuclear waste glass community, because it strives to ensure that vitrified products will be durable for thousands to tens of thousands of years. For the past three decades, this community has been attempting to fill this research gap by partnering with archeologists, museum curators, and geologists to identify hundred- to million-year-old glass analogues that have altered in environments (Figure 1) representative of those expected at potential nuclear waste disposal sites. Even with these partnerships, the process of identifying a waste glass relevant analogue is challenging; it requires scientists to relate data collected from short-term laboratory experiments to observations made from long-term analogues and extensive geochemical modeling.

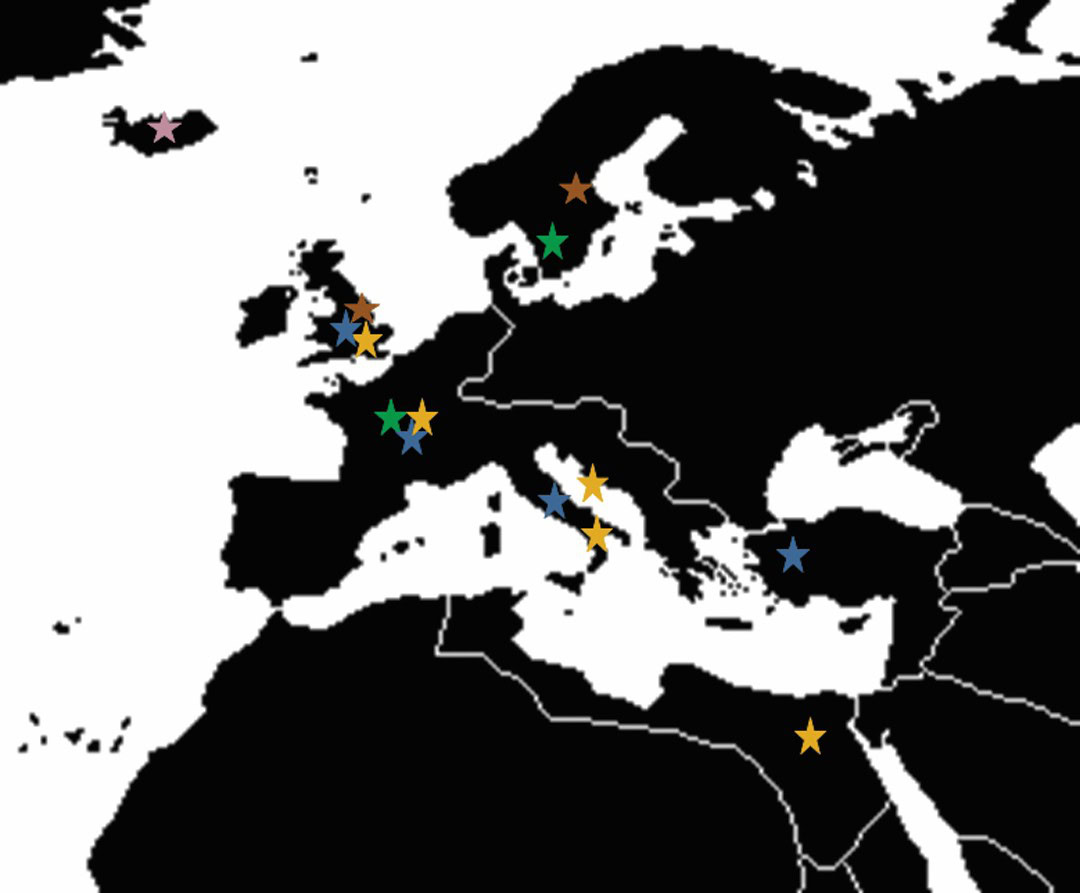
Figure 1. Locations and timeline of various types of excavated vitreous materials used as analogues to study durability of modern nuclear waste glasses. Credit: Jamie Weaver
Choosing an appropriate analogue: Initial considerations
When initially approaching the challenge of determining a glass alteration analogue, one could choose to limit the types of glasses to those that have compositions similar to nuclear waste glasses. Although it is very unlikely that an analogue will have exactly the same composition as a nuclear waste glass, it is important to study glasses that contain similar mass percentages of the baseline oxides of silica, aluminum, sodium, and, if possible, boron.
The difficulty of finding a one-to-one analogue is in part due to the complex elemental composition of nuclear waste. In general, the two most common types of vitrified nuclear wastes are low activity waste (LAW) and high-level waste (HLW). Classification of these wastes is most often based on present radionuclides and regulations regarding how these radioactive elements are immobilized and isolated from the environment. Additionally, researchers further distinguish glasses by their relative elemental concentrations.
For U.S.-based LAW glasses, baseline components are typically ~45 mass% SiO2 and ~20 mass% Na2O, mixed with ~6 mass% Al2O3, ~9 mass% B2O3, and ~20 mass% of other waste-derived oxides. Alternatively, U.S.-based HLW glasses contain ~30–55 mass% SiO2, ~15–20 mass% Na2O, ~4–22 mass% Al2O3, and ~5–20 mass% B2O3, with other oxides comprising the balance, either added in the frit or from the waste stream. HLW glasses also may contain more iron than LAW glasses. Depending on the origin of the waste stream—i.e., industrial waste from used nuclear fuel reprocessing or legacy defense nuclear waste—these other elements can vary widely in identity, concentration, and oxidation state.
Once a waste glass of interest is identified and its compositional range defined, then it is possible to begin looking for an appropriate analogue or set of analogues (Figure 1). An ideal analogue, as stated above, should contain most, if not all, of the main elements in the waste glass in mass% that fall within the composition range defined for the waste glass. Many previously studied analogues (see below) have two or three of the main elements of their corresponding waste glasses. In these cases, a second set of analogues that contains the relevant mass% of the other primary elements—and do not deviate more than a few mass% of the other elements—could be identified and studied. Combining both data sets could allow a researcher to develop a well rounded view of how major elements affect the long-term durability of waste glass.
Additionally, studying two or more analogues may provide insight into how a slight change in composition affects the overall durability of a sample. Understanding these interactions is important because glasses produced during vitrification often vary slightly from one melt batch to the next. These differences in composition are intentional and are designed to accommodate the special chemistry of a particular waste stream and to efficiently incorporate the waste elements into the glass.
Parallel to selecting an analogue that has a similar composition as the waste glass of interest, it also is important to choose an analogue that has been altered under relevant conditions. This is because how a glass alters can be partially determined by its altering environment.3 A candidate glass analogue should come from alteration environments similar to those that have been suggested or chosen for the disposal of nuclear waste glasses. Disposal areas are often designed to have engineered barriers, such as clay and granite, which will dictate chemistry of the environment in contact with the glass.
Analogues are samples with a past
In addition to the above considerations, scientists also collect information regarding the provenance, or history, of a possible glass analogue. They often mine this data from archeology, art conservation, and geology journals, or gather data through interviews with museum staff, geologists, or archaeologists who have studied the glass. Scientists can then use these findings to answer three subsequent questions about the analogue:4
- Is it ethical or feasible to analyze this glass?
- What ex-situ factors may have affected how this glass was altered, and what effects did they have?
- How does studying this glass help validate current glass alteration models?
Researchers should be aware of any cultural importance of a possible glass analogue and should carefully consider what physical and philosophical effects any analysis might have on an artifact’s long-term preservation. Researchers should ask whether analyzing the glass will change how it may be interpreted by future generations. It is important that researchers make these considerations with the advice of conservators, archeologists, art historians, and museum curators who specialize in the time period and geographical area associated with the artifact.
In all cases, an approved analogue should be analyzed using nondestructive methods if possible. However, if some destructive analysis is warranted to obtain critical information that otherwise could not be obtained, researchers must consider additional precautions. For instance, they should consider how destructive methods will alter the glass. They should be aware of what information they are removing from the object during analysis. They also need to consider what chemical changes the analysis technique might induce in the object, and whether this will impact its future preservation or interpretation.
Scientists studying the analogue also should consider how collected data can help validate or invalidate current glass alteration models. As stated in question three, a scientist should be aware of what part of the alteration model is being tested, which requires knowledge of glass corrosion science and theory.
Scientists should also consider what effect outside factors may have had on the alteration of glass analogues. Glass alteration models are developed from data collected on controlled experiments, but analogues are rarely altered under controlled conditions. Variability in alteration conditions could include annual changes of the pH of the groundwater that was in contact with the analogue, or fluctuations in microbial populations in the soil surrounding a buried analogue. It can be difficult to know all factors that might have influenced alteration of an ancient glass during its history, and it can be challenging to separate out the effects of each factor. However, simplified experiments performed in a laboratory setting can help disentangle these factors.
Data collected in the analysis of ancient glasses also can be utilized in preservation and historical interpretation of the artifact itself or similar pieces. For example, archeologists continue to attempt to gain an understanding of the methods used to make glass recovered at the Broborg site in Sweden (Figure 2 and images further below). Scientists hope that further analysis of ancient glasses may uncover redox conditions used by these ancient peoples and demonstrate a connection between iron working from that time and creation of the glasses.

Figure 2. Examples of hillfort glasses, which are manufactured glasses with compositions similar to some nuclear waste glasses. An amphibolite rock source for vitreous material, found at Broborg in Sweden, shows a molten top portion. Credit: John McCloy

Figure 2. Examples of hillfort glasses, which are manufactured glasses with compositions similar to some nuclear waste glasses. An amphibolite rock source for vitreous material, found at Broborg in Sweden, shows a molten top portion. Credit: John McCloy
Previous studies
Natural glasses
Most early studies of ancient glasses for the purpose of model validation have focused on alteration of natural glasses, such as basalts, tektites, and rhyolites.8–10 Rhyolitic glasses are high-silica natural volcanic glasses also is known as obsidian. These natural glasses range from hundreds of millions of years old (such as terrestrial, meteoritic, and lunar glasses) to only thousands of years old (some basaltic and rhyolitic glasses).
Tektites and rhyolitic glasses generally have high SiO2 concentrations (70–75 mass%), whereas basaltic glasses have lower SiO2 content (45–50 mass%). Unlike most nuclear waste glasses, natural glasses tend to contain low mass percentages of alkali elements. This low alkali content has made application of the alteration data collected on these natural glasses to most alkali-rich waste glasses difficult. However, even with these limitations, basaltic glasses have been used as fruitful analogues to some HLW glass formulations.11
Roman glasses
An excellent example of using anthropogenic glass analogues for modeling glass alterations can be found in the recent works of Verney-Carron and the Commissariat à l’ Énergie Atomique (CEA) on fractured, ~1,800-year-old glass blocks excavated in 2003 from a Roman shipwreck near the French island of Embiez.12 Many glasses—including nuclear waste glasses—fracture on cooling, and these fractures increase the reactive surface area of the glass. Because of the slow flow of water in and out of some cracks, increased surface area results in alteration rates in the cracks different from those on the exterior glass surfaces. The effects of these cracks on long-term alteration rates have been difficult to replicate in short-term laboratory tests because of the slow rate at which borosilicate glass alters.
In addition to showing that alteration mechanisms of archeological glass are similar to those described by a modern glass alteration model, the authors also discovered that alteration kinetics slightly varied depending on fracture location. They calculated a lower alteration rate for internal cracks that were not continually exposed to water as compared with exterior surfaces that were most likely in constant contact with water. Without renewal of fluid into cracks, the altering solution became saturated with dissolved glass elements, making it unfavorable for the glass matrix to undergo further dissolution.
In a recent follow-up to this study, Verney-Carron et al.13 used chemical modeling to simulate alteration of the glass in a variety of solutions, and they compared the results with reactive transport modeling of alterations in the cracks. They calculated that cracks inside the archaeological blocks had one to two orders of magnitude less alteration than the external surface because of strong coupling between rate of glass alteration and slow renewal of the internal crack altering solution. Additionally, narrow internal cracks precipitated crystals because of supersaturation of the solution in the crack with silicon, calcium, and aluminum. Formation of these crystals plugged the cracks, causing flow of water and alteration processes to halt.
On this side of the Atlantic, glass scientists at PNNL have been working with Italian archeologists to investigate the formation of alteration layers on second- to third-century CE Roman glass recovered from the Iulia Felix shipwreck.14 The Iulia Felix wreck is of particular interest, because its cargo included blue-green Roman glass fragments (Figure 3)—referred to in archeology literature as “naturally colored” glasses—and colorless and red shards, “artificially colored” by additions of reduced copper. Because ancient glassmakers usually added coloring agents in measured amounts to colorless glasses, artificially colored glass usually has a base composition similar to naturally colored Roman glasses. This slight modification makes it possible to investigate the effect of chemical composition on glass alteration within a set environment. Further research is currently underway on these artifacts.

Figure 3. Fragment of a Roman glass cup handle found in the Iulia Felix shipwreck, showing iridescent alteration layers. Credit: Denis Strachan, Joe Ryan
PNNL researchers have also focused on parts of these glasses that were encased in cemented sediments.14 The cementing material around the glass is magnesium-rich calcite, likely formed through corrosion of the glass in seawater saturated with naturally occurring dolomite and magnesium silicate precipitations. These magnesium silicate species form during alteration of some HLW glasses, and researchers identified similar species in the altering solution of nuclear waste glasses that were subjected to corrosion with a 1 mmol/kg MgCl2 solution.15,16 Both cited experiments were short term, but, when coupled with results from the Iulia Felix glasses, the researchers determined that, in undersaturated conditions, dissolution of the glass matrix is the main cause of formation of cementing phases. Burial materials in contact with glass are important in determining alteration conditions, and, if possible, fragments should be collected with sediments in place (Figure 4).

Figure 4. Glass collected in-situ with contacted soil at the Aquileia site in Italy. Aquileia was a major glass-producing center in Roman times. Credit: Denis Strachan
Iron slag
Many scientists have gained insight from other ancient vitreous materials in addition to glasses. A few studies in recent years have focused on alteration of metallurgical slags—glassy materials produced when metals are smelted from ore.17 Slag is comprised of a mixture of metal oxides, typically calcium oxide, and silica. Iron blast furnace slags, for instance, are formed from silica in the iron ore and added limestone or a similar calcium source. Similar vitreous materials may form when clays from furnaces are heated to high temperatures when processing iron (Figure 5). Aged slags produced during ironworking represent an alteration condition in which silica-rich materials are altered in the presence of metallic iron. This situation is similar to what may happen to nuclear waste glass as it ages in stainless-steel containers.
Researchers identified such altered slags from the 16th century CE and recovered from an excavated blast furnace site located in Normandy, France. Geological conditions of the burial (saturated clay) are similar to those described for disposal of some waste glasses in France. French scientists at the CEA have studied these samples using short-term wet laboratory experiments as well as analysis of long-term alteration layers on the artifact surfaces.17

Figure 5. Scientific collaboration at work in Uppsala, Sweden, showing investigation of vitrified iron furnace walls. Shown (left to right) are John McCloy, glass scientist of Washington State University; Eva Hjärthner-Holdar, archaeometallurgist in Arkeologerna, Geoarkeologiskt Laboratorium; and Rolf Sjöblom, geochemist with Tekedo and Luleå University of Technology. Credit: Jamie Weaver
Researchers designed wet laboratory experiments to closely reproduce the proposed French nuclear waste disposal settings. They covered SON68, a nonradioactive version of a French HLW glass, wih an iron–steel composite and injected it with a synthetic solution at a flow rate chosen to test the glass under diffusion-controlled conditions. The researchers also used this alteration routine on synthetic archeological slags that had the same composition as excavated samples. They determined significant similarities in the increase of glass alteration in the presence of iron from comparison of the two altered samples. This result supports previous studies that have shown a deleterious effect of reduced iron, which results in an increased rate of glass dissolution, an important result that researchers can use to aid modeling of long-term durability of glasses stored in iron-based containers.18
Swedish hillfort glasses
Researchers recently have identified glasses that were produced almost 1,500 years ago on a hillfort in Sweden that have been altered by surface atmospheric conditions, which have analogies to the near-surface disposal planned for some low-level nuclear waste glasses, including those at Hanford.19 A hillfort is a building structure used as a fortified refuge, trading post, or defended settlement, where an elevated location relative to the surrounding area provides a defensive advantage.20 There are many hillforts in Europe, and these fortifications usually follow the contours of a hill, consist of one or more lines of earthworks (including stockades or defensive walls), and are surrounded by external ditches. More than 100 of the European hillforts, most from the Iron Age, are vitrified, and much controversy continues to surround how this occurred (see bonus content: “Broborg hillfort case study“).20
A selection of glasses from the Broborg hillfort site in Sweden contain iron at concentrations between those detected in basaltic and Roman glasses. Similar to basaltic glasses, some of the Broborg glass samples contain embedded crystalline phases, such as magnetite and other spinels. The current acceptance criteria for Hanford LAW glasses allow up to a few vol% of spinel crystals. The hillfort vitrified material remains exposed today to surface weathering cycles or, in some cases, is shallowly buried by local soil.7 Although research on these ancient vitreous materials remains in its early stages, WSU researchers have made promising headway in an attempt to synthetically reproduce these glasses. In addition, PNNL researchers have been working with Swedish archeologists, scientists, and state officials to acquire fresh near-surface altered samples from the Broborg site.
Medieval glasses
It is sometimes helpful to analyze a glass that has been altered under two or more conditions simultaneously. In these cases, the focus of understanding is placed on whether a glass alteration model can be extrapolated to include multiple altering conditions. Alteration data from several environments can be extremely helpful when attempting to decide what disposal setting and geology is best for a specific type of waste glass. Researchers have discovered medieval stained and nonstained glass windows to be ideal analogues for these studies, because the windows often have undergone alteration under two or more settings21—glass window panels facing outward are exposed to an alteration environment different from those facing inward. A few rare glass samples that have been found in architectural structures have also been excavated from groundwater-saturated soils,21 thus providing a third environment to compare alteration data.
Because builders, craftsmen, and church officials kept excellent records, medieval glass-manufacturing methods of most stained glass windows are well-known. Therefore, glass scientists can identify conditions under which the glasses were made. In most cases, craftsmen made medieval glasses in either soda (sodium-rich) or potash (potassium-rich) compositions through melts of a blend of washed siliceous sand and a flux.22 The flux material was either plant-based (beech or fern ash) or mineral-based (natron) and, when added to other glass-forming materials, lowered the melting temperature.
Glassmakers achieved color either by adding metals or oxides of cobalt, chromium, manganese, copper, or iron and controlling redox conditions of the melt, or by applying a thin layer of a previously colored glass to the surface of a clear glass. Minor components in these medieval glasses are thus transition metals that are relevant to some nuclear waste glasses. Chemical durability different from the sodium-rich versus potassium-rich glasses as well as presence of multiple compositions with various transition metals provide a large dataset for studying composition-dependent alterations as well.
Scanning electron microscopy studies on medieval stained glasses from England, Italy, and France have revealed that the type of weathering has an effect on thickness and structure of the alteration layer(s). Potash glasses exposed to an exterior environment produce thick, uneven alteration layers with some pitting, along with pervasive microcracking.23 In contrast, potash glasses that have been buried and exposed to groundwater have nearly even alteration layers with little to no cracking.
Potential future glasses for study
There are many less-well-studied ancient glasses from around the world that deviate significantly in composition from the glasses described above. These glasses could provide researchers with new insights into the effects of compositional and environmental differences on glass alteration. These include high-alumina glasses from India and some regions in Turkey, a special subset of Byzantine glasses that contain a moderate level of alumina plus some boron,24 and certain Chinese glasses with high barium oxide content. Researchers currently are considering some of these glasses as possible analogues, although their rarity makes them challenging to acquire for study.
Researchers also need to identify high-concentration boron (~10 mass% B2O3) aluminosilicate glasses that have been altered in waste-disposal-relevant environments. Boron is a primary component in all waste glasses and has increased, in certain cases, the durability of silicate glasses.25
Boron made its mark on the glass industry in the early 1900s, when Corning patented a series of boron-containing silicate glasses that could withstand temperature shock—Pyrex.26 At this time, there were many recorded accidents related to breakage of hot glass railway lanterns during cold rainstorms. Glass breakage caused the railway signals to fail, catching innocent people unaware of oncoming trains. Replacing soda in the glass with boron oxide solved the glasses’ thermal shock problem.
Today, we find borosilicate glasses in almost every home, office space, and laboratory. In addition, commercial uses have utilized the poor durability of some boron-containing glasses. One example is Vycor, in which a phase-separated sodium borosilicate glass is selectively leached of its sodium borate phase, leaving a skeletal silicate network that is usually further consolidated to produce a low-cost nearly pure silica. Clearly, chemical durability concerns are relevant to ancient materials and to today’s and likely tomorrow’s commercial glass processing.
Challenges remain
The studies reviewed above are only a portion of those conducted to date on ancient glasses.27,28 However, their relevance to the validation of glass alteration models is significant. There are many challenges associated with analyzing ancient glasses for this purpose. However, the payoff of having a composition- and environment-dependent model of glass alteration validated by many data points, spanning days to millions of years, will be momentous. A better understanding of long-term glass alteration will allow for a more accurate prediction of the long-term performance of vitrified nuclear waste and could aid continued formulation of waste glasses that will be durable for “thousands of years”.2 In the process, knowledge gained on glass alteration can help create industrial glasses with engineered durability as well as help museums and historical societies in the preservation of cultural heritage objects.
Read more: “Broborg hillfort case study“
Capsule summary
The challenge
Studying how glass alters over time, especially large spans of time, is a challenging research problem. However, thorough understanding of these mechanisms is critical for prediction and design of glasses that can safely store nuclear waste for long periods of time.
What’s old is new again
Ancient artifacts represent a rich source of material to study long-term glass alterations under various environmental and exposure conditions. Although artifacts often have compositions different from new glasses, they offer many potential analogues for study.
Future models
Better understanding of long-term glass alteration allows more accurate prediction of the performance of vitrified nuclear waste to help develop waste glasses that will be durable for many millenia.
Cite this article
J. L. Weaver, J. S. McCloy, J. V. Ryan, and A. A. Kruger, “Ensuring longevity: Ancient glasses help predict durability of vitrified nuclear waste,” Am. Ceram. Soc. Bull. 2016, 95(4): 18–23.
About the Author(s)
Jamie Weaver is a Ph.D. student in the Department of Chemistry at Washington State University (Pullman, Wash.) and a Ph.D. intern at PNNL. Joseph Ryan is with the Energy and Environment Directorate at Pacific Northwest National Laboratory (Richland, Wash.). John McCloy is associate professor in the School of Mechanical and Materials Engineering at Washington State University and a joint appointee with Pacific Northwest National Laboratory. Albert Kruger is with the Office of River Protection at the Department of Energy (Richland, Wash.).
Issue
Category
- Art, archeology, and conservation science
- Energy materials and systems
- Glass and optical materials
Article References
1R.H. Brill, “The record of time in weathered glass,” Archaeology, 14 [1] 18–22 (1961).
2C.M. Jantzen, K.G. Brown, and J.B. Pickett, “Durable glass for thousands of years,” Int. J. Appl. Glass Sci., 1 [1] 38–62 (2010).
3B. Dal Bianco, R. Bertoncello, L. Milanese, and S. Barison, “Glasses on the seabed: Surface study of chemical corrosion in sunken Roman glasses,” J. Non-Cryst. Solids, 343 [1–3] 91–100 (2004).
4W.M. Miller, N. Chapman, I. McKinley, R. Alexander, and J.A.T. Smellie, Eds., Natural analogue studies in the geological disposal of radioactive wastes. Elsevier, New York, 2011.
5E. Youngblood, “Celtic vitrified forts: Implications of a chemical-petrological study of glasses and source rocks,” J. Archaeol. Sci., 5 [2] 99–121 (1978).
6P. Kresten and B. Ambrosiani, “Swedish vitrified forts–A reconnaissance study,” Fornvännen, 87, 1–17 (1992).
7P. Kresten, L. Kero, and J. Chyssler, “Geology of the vitrified hill-fort Broborg in Uppland, Sweden,” GFF, 115 [1] 13–24 (1993).
8J. Crovisier, H. Atassia, V. Dauxa, J. Honnoreza, J. C. Petita, and J. P. Eberhart, “A new insight into the nature of the leached layers formed on basaltic glasses in relation to the choice of constraints for long term modelling,” MRS Proceedings 127. Cambridge University Press, Cambridge, U.K., (1988).
9I. Techer, T. Advocat, J. Lancelot, and J.-M. Liotard, “Basaltic glass: Alteration mechanisms and analogy with nuclear waste glasses,” J. Nucl. Mater., 282 [1] 40–46 (2000).
10M.C. Magonthier, J.C. Petit, and J.C. Dran, “Rhyolitic glasses as natural analogues of nuclear waste glasses: Behaviour of an Icelandic glass upon natural aqueous corrosion,” Appl. Geochem., 7 [Supplement 1] 83–93 (1992).
11B. Parruzot, P. Jollivet, D. Rebiscoul, and S. Gin, “Long-term alteration of basaltic glass: Mechanisms and rates,” Geochim.Cosmochim. Acta, 154, 28–48 (2015).
12A. Verney-Carron, S. Gin, and G. Libourel, “A fractured Roman glass block altered for 1800 years in seawater: Analogy with nuclear waste glass in a deep geological repository,” Geochim. Cosmochim. Acta, 72 [22] 5372–85 (2008).
13A. Verney-Carron, S. Gin, P. Frugier, and G. Libourel, “Long-term modeling of alteration-transport coupling: Application to a fractured Roman glass,” Geochim. Cosmochim. Acta, 74 [8] 2291–315 (2010).
14D.M. Strachan, J.V. Crum, J.V. Ryan, and A. Silvestri, “Characterization and modeling of the cemented sediment surrounding the Iulia Felix glass,” Appl. Geochem., 41, 107–14 (2014).
15B. Grambow and D. Strachan, “Leach testing of waste glasses under near-saturation conditions”; pp. 623–34 in Scientific basis for nuclear waste management VII. North-Holland, New-York, 1984.
16T. Maeda, H. Ohmori, S. Mitsui, and T. Banba, “Corrosion behavior of simulated HLW glass in the presence of magnesium ion,” Int. J. Corros., 2011, 796457 (2011).
17A. Michelin, E. Leroy, D. Neff, J.J. Dynes, P. Dillmann, and S. Gin, “Archeological slag from Glinet: An example of silicate glass altered in an anoxic iron-rich environment,” Chem. Geol., 413, 28–43 (2015).
18E. Burger, D. Rebiscoul, F. Bruguier, M. Jublot, J.E. Lartigue, and S. Gin, “Impact of iron on nuclear glass alteration in geological repository conditions: A multiscale approach,” Appl. Geochem., 31, 159–70 (2013).
19R. Sjöblom, H. Ecke, and E. Brännvall, “Vitrified forts as anthropogenic analogues for assessment of long-term stability of vitrified waste in natural environments,” Int. J. Sustain. Dev. Plann., 2013.
20D. Harding, Iron age hillforts in Britain and beyond. Oxford University Press, Oxford, U.K., (2012).
21J. Sterpenich and G. Libourel, “Using stained glass windows to understand the durability of toxic waste matrices,” Chem. Geol., 174 [1] 181–93 (2001).
22J. Henderson, “The raw materials of early glass production,” Oxford J. Archaeol., 4 [3] 267–91 (1985).
23T. Lombardo, L. Gentaz, A. Verney-Carron, A. Chabas, C. Loisel, D. Neff, and E. Leroy, “Characterisation of complex alteration layers in medieval glasses,” Corros. Sci., 72, 10–19 (2013).
24T. Rehren, P. Connolly, N. Schibille, and H. Schwarzer, “Changes in glass consumption in Pergamon (Turkey) from Hellenistic to late Byzantine and Islamic times,” J. Archaeol. Sci., 55, 266–79 (2015).
25R.W. Revie and H.H. Uhlig, Uhlig’s corrosion handbook, Vol. 51. Wiley, New York, 2011.
26W.B. Jensen, “The origin of pyrex,” J. Chem. Educ., 83 [5] 692 (2006).
27R.H. Brill, Chemical analyses of early glasses, Vol. 2: Tables. The Corning Museum of Glass, Corning, New York, N.Y. (1999).
28J. Henderson, Ancient glass: An interdisciplinary exploration. Cambridge University Press, Cambridge, U.K. (2013.
Related Articles
Bulletin Features
Ferroelectrics for energy-efficient electronics
It has always fascinated me how fast computers have improved over the past decades. It is still hard to grasp that a modern smartphone has more processing power than all computers existing in the 1960s combined—while running on a tiny battery. How was this dramatic improvement of energy efficiency possible?…
Market Insights
The evolving discipline of demand forecasting can improve competitiveness and control costs
What does the future hold? It is a question that everyone has contemplated at one time or another, and a mystery that has obsessed writers, mystics, philosophers, clerics, astrologers, and others for centuries. For businesses, the question is more than philosophical. Discovering clues to what is next is essential to…
Bulletin Features
Sustainable ceramics production: Environmental considerations in tile manufacturing
Ceramics are often regarded as a sustainable material choice thanks to their high durability and chemical stability, which enable a long service life. But ceramics come with environmental baggage in other areas, such as raw material sourcing, processing, and disposal. The specifics of this baggage are unique to each ceramic…





